Traditional oil-based drilling muds (OBMs) have a relatively high solid content, which is detrimental to penetration rate increase and reservoir protection. Aimed at solving this problem, an organoclay-free OBM system was studied, the synthesis methods and functioning mechanism of key additives were introduced, and performance evaluation of the system was performed. The rheology modifier was prepared by reacting a dimer fatty acid with diethanolamine, the primary emulsifier was made by oxidation and addition reaction of fatty acids, the secondary emulsifier was made by amidation of a fatty acid, and finally the fluid loss additive of water-soluble acrylic resin was synthesized by introducing acrylic acid into styrene/butyl acrylate polymerization. The rheology modifier could enhance the attraction between droplets, particles in the emulsion via intermolecular hydrogen bonding and improve the shear stress by forming a three-dimensional network structure in the emulsion. Lab experimental results show that the organoclay-free OBM could tolerate temperatures up to 220 °C and HTHP filtration is less than 5 mL. Compared with the traditional OBMs, the organoclay-free OBM has low plastic viscosity, high shear stress, high ratio of dynamic shear force to plastic viscosity and high permeability recovery, which are beneficial to penetration rate increase, hole cleaning and reservoir protection.
Traditional oil-based drilling muds (OBMs) use organoclay[1] as thickening agents and blown asphalt or organic lignite to reduce fluid loss, which have played an important role in the development of drilling engineering and are still widely used today in drilling complex, deep and ultra-deep formations and complex structural wells. However, traditional OBMs have some limitations. For example, colloidal particles of organoclay and blown asphalt have negative influence on the rate of penetration[2, 3]. After aging, organoclay and blown asphalt are likely to cause the drilling fluids to thicken. Colloidal particles can become pollutants in reservoirs. The drilling fluid is difficult to increase to high density at high colloidal content. Thus, OBMs with low colloidal content are widely used[4]. However, low colloidal content reduces the shear stress of OBMs, resulting in a poor cleaning effect[5, 6, 7].
Organoclay-free OBM has the following advantages compared to traditional OBMs: excellent rheological properties, unique shear stress structure, strong thixotropy[6, 8] and good shear shinning properties; Organoclay-free OBM does not use colloidal components such as organoclay and asphalts, enabling a high rate of penetration and a good reservoirs protection performance. Therefore, organoclay-free OBM is beneficial for efficient exploitation of oil and gas resources and has become an important direction of drilling fluid development in the future.
The study of organoclay-free OBM systems and additives have been undertaken for some time worldwide, but only recently in China. The organoclay-free OBMs, developed by Halliburton and other companies, have superior rheology and reservoir protection performance and good field application effects[9]. However, the functional mechanism of organoclay-free OBM is not well understood, and there are only a few studies reporting the additives, rheology modification mechanism of the organoclay-free OBM and the drilling fluid system. An organoclay-free OBM is described in this study, the synthesis of key additives, functional mechanism and system performance are also presented.
Key additives of organoclay-free OBM include: rheological modifier (RM), primary and secondary emulsifiers, and filtrate reducer. Among them, RM is the main additive.
1.1.1. Synthesis of RM
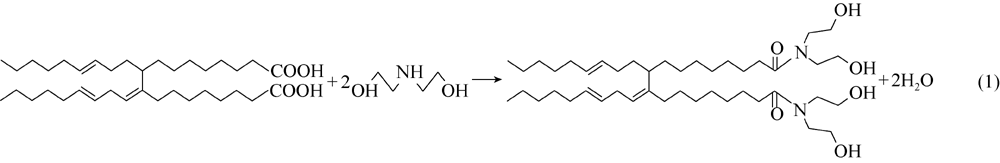
The RM is high in viscosity due to the intermolecular hydrogen bonding between fatty acid dimers and suitable to be used as the rheological modifier for OBMs. However, RM has poor hydrophilicity. In this study, diethanolamine was used for the amidation of RM to increase its hydrophilicity. The reaction is shown in equation (1). The hydrophilicity of the fatty acid dimer is expected to increase with the addition of amide groups according to hydrophile-lipophile balance (HLB) theory. Theoretically, hydrophilicity of the fatty acid dimer can be also enhanced using saponification of the fatty acid with a strong base. However, the internal phase of the OBM generally is high concentration brine, which reduces the hydrophilic effect of the fatty acid salt. The HLB was estimated by Davies base method in this study. HLB base values of the hydrophilic functional groups— COOK+, — COONa+, R3N(tertiary amine), — COOH, — OH and — O— are 21.1, 19.1, 9.4, 2.1, 1.9 and 1.3, respectively[10].
Detailed synthesis process of RM is: fatty acid dimer and diethanolamine were mixed at the ratio of 1:2.05 (molecular fraction) and placed into a four-necked flask. An electric mixer, condenser and reflux apparatus and dean stark trap were attached to the flask and nitrogen was injected into the mixture for 10 min before the reaction was started. The mixture was heated by oil bath quickly to the reaction temperature of 175 ° C to react for 4 hours. During the reaction process, water in the dean stark trap was removed in time. At the end of the reaction, the brown thick fluid produced was the RM.
1.1.2. Effects of RM dosage on emulsion modulus
An emulsion with an oil-water ratio of 85:15 (5# white oil: water) and 3% (wt/V) of emulsifier SPAN-80 was prepared using a shearing mixer. Dynamic frequency of the emulsion was scanned using a Haake MARS III rheometer in cone plate mode to test the storage modulus (G') and loss modulus (G'') of the emulsion. The frequency scanning range was 0.1 to 10 s-1 and test temperature was held at 25 ° C and shear stress was maintained at a constant 0.1 Pa.
Experimental results are shown in Fig. 1. It can be seen pure emulsion has a higher G'' compared to G' within the frequency range used in this study. This means the emulsion mainly has viscous deformation and is in liquid state. After RM is added, G' is higher than G'', indicating that the emulsion would change from a liquid state into a “ gel state” . When the RM concentration is lower than 3 g/L, G' increases with the increase of RM concentration, but reduces when the concentration of RM is greater than 3 g/L. This could be because the interaction force between the emulsion droplets is overly large, leading to unstable flocculation of the emulsion and reduction of G'.
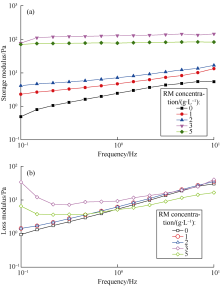 | Fig. 1. Effect of rheological modifier (RM) concentration on storage modulus (a) and loss modulus (b) of the emulsion. |
It is reported in the literature [11] that, with the increase of structural force in an emulsion, the influence of frequency on G' decreases over time, i.e., the slope of a G' curve decreases. It can be seen in Fig. 1(a) that the slope of the G' curve decreases gradually with the increase of RM content. This indicates that the interaction force in the emulsion increases with the increase of RM concentration.
1.1.3. Functional mechanism of RM
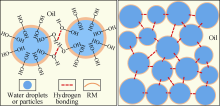
The functional mechanism of RM in organoclay-free OBM is shown in Fig. 2. Stable emulsion can be formed in the OBM, thanks to the synergetic effect of the primary and secondary emulsifiers. With an amphiphilic structure, RM can adsorb onto the oil-water interface. Due to the solubilization of the emulsifier on the oil-water interface, the concentration of RM increases at the oil-water interface. Due to hydrogen bonding between the RM molecules, RM would form a “ film” at the oil-water interface. On one hand, RM accumulation on the interface strengthens the interface film, which is conducive to the stability of the emulsion. On the other hand, the hydrogen bonding between RM adsorbed on different water droplets enhances the droplet-droplet interaction force in the emulsion.
There are hydroxy groups on particle surfaces, e.g. waterborne acrylic resin filtrate reducer, calcium carbonate superfine powder and barite. In drilling fluid, RM can not only adhere to the oil-water interface, but also the solid-liquid interface due to the hydrogen bonding effect. This can strengthen droplet-droplet, droplet-particle and particle-particle interactions, thus forming a 3D network structure connected by weak interactions in the emulsion (weak gel structure)[8], enhancing the structural force of the emulsion to serve as shear stress.
Within the appropriate concentration (0-3 g/L), RM can increase emulsion structural force and stability. However, flocculation occurs when the RM concentration is too high, which reduces emulsion stability and emulsion structural force.
1.2.1. Design principle of emulsifier resistant to high temperatures
Appropriate HLB value: Emulsifiers are the base to maintain emulsion stability and improve other properties of OBM. To form stable oil-in-water emulsion, the emulsifier must be hydrophilic and lipophilic, and be hydrophilic and lipophilic balance under certain conditions. For nonionic emulsifiers, the HLB value generally ranges between 3.0 and 6.0. The performance of an ionic emulsifier is highly sensitive to water salinity. For ionic emulsifiers with specific HLB values, the hydrophilic interaction of the emulsifier decreases gradually with the increase of water salinity. In OBM, two different emulsifiers (primary and secondary emulsifiers) are generally used to form the dense composite membrane[12] to achieve the best emulsification effect. The primary emulsifier is generally a fatty acid or modified fatty acid with a lower HLB value. The secondary emulsifier has a higher HLB value, which can stabilize the emulsion and also wet the solid phase.
Strong interaction forces at the lipophilic and hydrophilic ends: the molecular structure of the emulsifier must tolerate high temperatures. In addition, the emulsifier should have strong interactions with both oil and water phases at the interface. Therefore, high-temperature stability requires strong hydrophilic and lipophilic groups (generally hydrophobic groups of similar carbon chain length as the oil phase) in emulsifiers.
1.2.2. Primary emulsifier
In this study, the primary emulsifier was obtained from the reaction of fatty acid and maleic anhydride. The synthesis method is reported in reference [13]. The hydrophilic groups are carboxylic acids and the hydrophobic groups are long chain fatty acids. The bivalent soap salt generated by the reaction between the carboxylic acid groups and CaO in the OBM not only has strong hydrophilic property but also increases emulsion stability based on the directional wedge theory[14].
1.2.3. Secondary emulsifier
The secondary emulsifier in this drilling fluid is a fatty acid amide. The hydrophilic group is the amide group and the hydrophobic group is the long chain fatty acid. Its synthesis principles are: Step 1: partial amidation products are generated through the amidation of fatty acid polyamines and fatty acids. Step 2: the partial amidation products react with diacid to amidate the primary amine groups completely and generate the secondary emulsifier of fatty acid amides.
1.2.4. Evaluation of high temperature emulsion performance
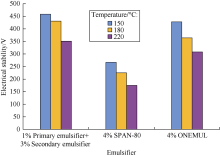
The following OBM was prepared: base fluid of 5# white oil and 30% CaCl2 aqueous solution at the ratio of 85:15 + 1% organoclay + 1% primary emulsifier + 3% secondary emulsifier + 3% CaO. The OBM was stirred at a rate of 5 000 r/min for 30 min. The prepared OBM was placed into a roller furnace for high-temperature aging. The roller furnace temperatures were set at 150 ° C, 180 ° C and 220 ° C respectively. At the end of the aging process, the mixture was stirred for 2 min at a rate of 5 000 r/min. An electrical stability tester was used to test the electrical stability (ES) of the drilling fluid. Fig. 3 shows the emulsion formed using the primary and secondary emulsifiers has a high ES after high-temperature aging. It is superior to SPAN-80 and ONEMUL in high-temperature emulsification performance.
1.3.1. Synthesis of filtrate reducer
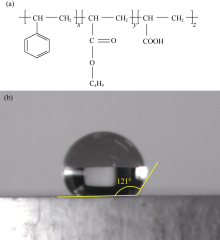
Under high temperature conditions, without filtrate reducers like organoclay, asphalt or lignite, it would be difficult to reduce fluid loss. But modified asphalt and organoclay are lipophilic colloids, which would increase the viscosity of the drilling fluid, and thus reduce rate of penetration. A waterborne acrylic resin with strong hydrophilic property has been developed by introducing hydrophilic monomer acrylic acid into the emulsion polymerization of styrene and butyl acrylate. The chemical structure is shown in Fig. 4a. The water-phase contact angle of the pure styrene acrylic resin is relatively large. However, the contact angle of distilled water on the developed waterborne acrylic resin pressed pill decreases to 121° (Fig. 4b). The lipophilicity of waterborne acrylic resin is stronger than the hydrophilic property and the majority of the resin particles in the emulsion is in the oil phase, resulting in the bending of the oil-water interface. This increases emulsion stability[15]. The glass transition temperature of the developed waterborne acrylic resin is 103 ° C. When the ambient temperature is higher the glass transition temperature, the waterborne acrylic resin is in the rubber state, which is beneficial to plugging of the formation pores. In addition, the waterborne acrylic resin type materials generally have a film-forming property, which is conducive to the formation of compact filter cake to reduce fluid loss.
1.3.2. High-temperature performance of the fluid loss reducer
The following drilling fluid was prepared to evaluate the performance of the filtrate reducer: base fluid of 5# white oil and 30% CaCl2 aqueous solution at the ratio of 85:15+ 1% organoclay + 1% primary emulsifier + 3% secondary emulsifier + 3% CaO + barite (ρ = 1.5 g/cm3).
API filter loss (FLAPI) and high-temperature high-pressure (HTHP) filter loss (FLHTHP) of the drilling fluid after aging at the high temperature of 220 ° C for 16h are shown in Table 1. The HTHP filter loss was measured at 180 ° C and 3.5 MPa. Apparently, the commonly used fluid loss reducers like oxidized asphalt and organic lignite aren’ t suitable for the organoclay-free environment with high filtration loss, failing to meet the requirements of drilling engineering. The synthesized polymer fluid loss reducer presented in this paper has excellent filtration reduction performance, with a HTHP filtration loss of less than 5 mL.
| Table 1 Comparison of performance of different filtrate reducers for OBM. |
The 5#white oil wa taken as the base oil. Firstly, the 5# white oil has high flash and ignition points, which can ensure the safety of the drilling process. Secondly, the aniline point of the base oil should be higher than 60 ° C under general conditions, and the 5# white oil has a high aniline point which can cause little damage to the rubber parts of the drilling machinery. Thirdly, the 5# white oil has appropriate viscosity, which is beneficial for rheology control. Fourthly, the 5# white oil has low toxicity, making it environmentally friendly.
The wetting agent is used to reverse the wettability of barite surface, to change the hydrophilic surface into a lipophilic surface, and thus enhance dispersion of barite in the OBM. The wetting agent can influence the filter loss of OBM considerably. With poor wettability, barite can easily precipitate, and the weighted drilling fluid has high filter loss. Lecithin is a natural surfactant, with HLB ranging between 7.5 and 8.0[16]. It has excellent emulsification, scattering and wetting properties. Therefore, lecithin was chosen as the wetting agent.
To block the formation pores of different sizes, two kinds of superfine calcium carbonate powders with different sizes (25 μ m (500-mesh) and 6.5 μ m (2 000-mesh)) were used as plugging materials.
Under the same density, high-purity barite has a small impact on the viscosity of the drilling fluid, so in this study, high-purity barite with a density of 4.3 g/cm3was used as the weighting agent.
The following organoclay-free OBM formula was formed through lab tests: base fluid (the volume ratio of 5# white oil to 30% CaCl2 aqueous solution is 85:15) + 0.3% RM + 2 % primary emulsifier + 3% secondary emulsifier + 1.5% wetting agent + 2% waterborne acrylic resin + 4% calcium carbonate superfine powder (25 μ m) + 4% calcium carbonate superfine powder (6.5 μ m) + 4% CaO + barite (ρ = 2.2 g/cm3).
The rheological property, filtration property and reservoir protection property of the organoclay-free OBM were tested, and compared with those of traditional OBM with organoclay.
The traditional OBM formula used was: base fluid (with the volume ratio of 5# white oil to 30% CaCl2 aqueous solution of 85:15) + 1.0% VGPLUS (organoclay) + 5% ONEMUL (emulsifier) +1.5% VERSACOAT (wetting agent) + 4% VERSATROL (fluid loss reducer) + 0.5% SACKBLACK (fluid loss reducer) + 4% CaO + barite (ρ = 2.2 g/cm3).
2.2.1. Evaluation of general properties
Table 2 shows the general properties of the organoclay-free OBM and the traditional OBM before and after aging. The aging was conducted at 220 ° C for 16 h. HTHP filter loss was tested under 180 ° C and 3.5 MPa. Rheological performance was tested under 50 ° C. It can be seen that both OBMs can tolerate high temperatures up to 220 ° C, have ES of greater than 400 V after aging and HTHP filter losses within the acceptable range. However, compared with the traditional OBM, the organoclay-free OBM has the following advantages: under the same density, organoclay-free OBM containing no colloidal solid phase has lower viscosity, which is good for increasing the rate of penetration. The traditional OBM has high viscosity before and after aging, which has negative effect on increasing rate of penetration. Besides, the organoclay-free OBM has higher yield point, gel strength and higher ratio of yield point to plastic viscosity before and after aging than traditional OBMs, which is conducive to cutting-carrying.
| Table 2 Comparison of organoclay-free OBMs and traditional OBMs. |
2.2.2. Reservoir protection performance
Artificial cores 25 mm in diameter and 50 mm long with the gas permeability of approximately 50 - 100 × 10-3 µ m2 were used to test changes of gas permeability of the cores before and after polluted by the organoclay-free OBM and the traditional OBM and compare their performance in reservoir protection. Firstly, the initial gas permeability (K0) of the cores was tested. Secondly, one surface of the man-made core was polluted for 2 h at 60 ° C and 3.5 MPa using a HTHP drilling fluid dynamic damage device (JHMD-II type, Jinzhou Modern Petroleum Scientific Technology Company). Thirdly, the polluted surface was cut and the reverse gas permeability of the polluted core (K1) was tested. Finally, the permeability recovery was calculated using the experimental results. Three parallel tests were carried out and the mean permeability recovery was calculated.
As shown in Table 3, the mean permeability recovery of cores polluted by the organoclay-free OBM system is greater than 95%, while permeability recovery of the traditional OBM is lower than 92%. Therefore, although the traditional OBM has fairly good reservoir protection performance, the organoclay-free OBM without organoclay and asphalt colloids can further reduce damage to the formation and has a better reservoir protection performance.
| Table 3 Comparison of permeability recovery of cores polluted by the organoclay-free OBM and the traditional OBM. |
In this study, a kind of organoclay-free OBM system has been developed, which contains four key additives (RM, primary emulsifier, secondary emulsifier and filtrate reducer). RM is a modified fatty acid that could replace organoclay in OBMs, which is prepared by the reaction between dimer acid and diethanolamine. After RM is added, the storage modulus of the emulsion increases significantly and the emulsion is in a “ gel state” . The primary emulsifier is a modified fatty acid and the secondary emulsifier is a fatty acid amide. Both primary and secondary emulsifiers have strong hydrophilic and lipophilic groups, and excellent emulsion performance after high-temperature aging, superior to similar commercial products made by foreign companies. The filtrate reducer is a kind of waterborne acrylic resin with good emulsion stabilizing and fluid loss reducing performance. It can control the HTHP filter loss in organoclay-free OBMs at less than 5 mL.
RM is the main additive. The functional mechanism of RM: In organoclay-free OBMs, RM is adsorbed on the oil-water interface and solid-liquid surface, strengthening the droplet-droplet, droplet-particle and particle-particle interactions in the emulsion through hydrogen bonding, forming a 3D network structure connected by weak interactions, thus strengthening the gel structure of the emulsion, improving the rheology and stabilizing the performance of the emulsion.
Performance evaluation experiments show that compared with the traditional OBMs, the organoclay-free OBM has low plastic viscosity, high shear stress, high ratio of yield point to plastic viscosity and high permeability recovery, which are beneficial to penetration rate increase, hole cleaning and reservoir protection.
The authors have declared that no competing interests exist.
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|