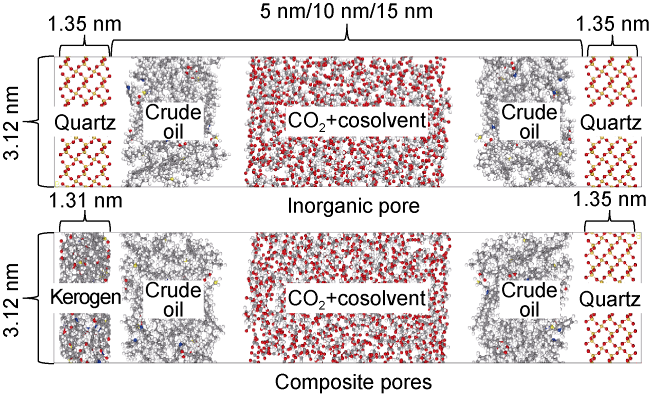
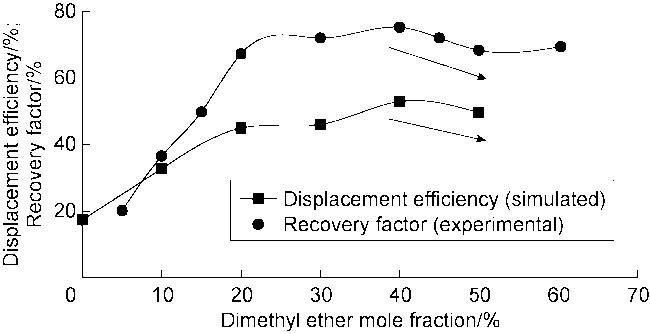
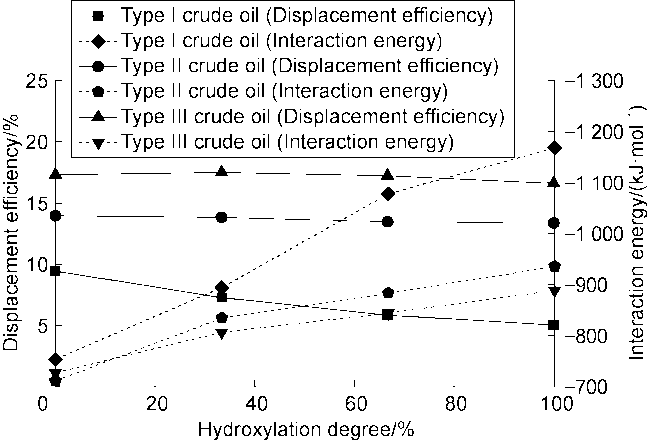
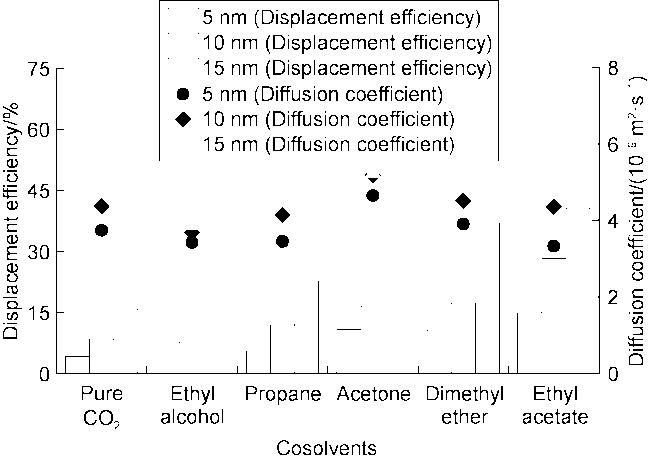
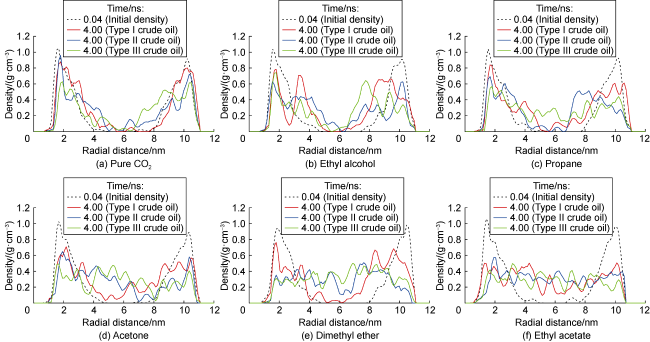
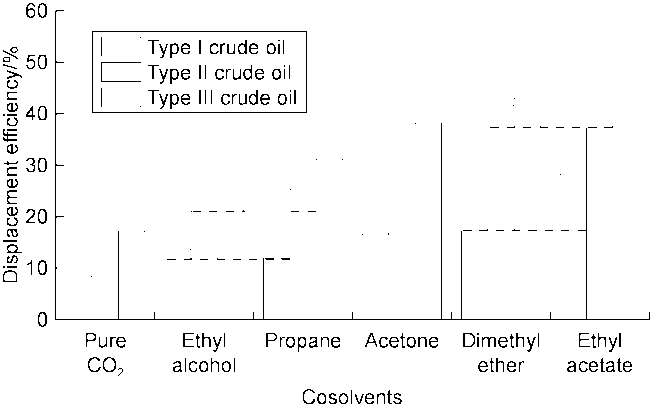
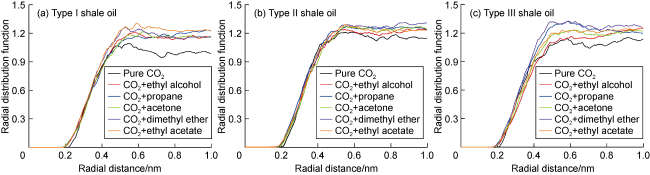
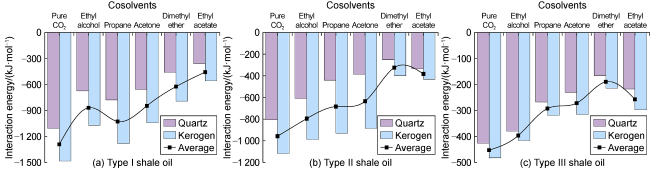
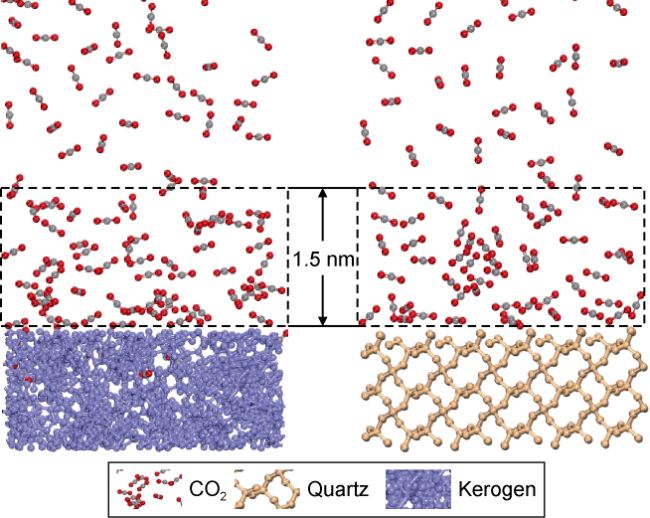
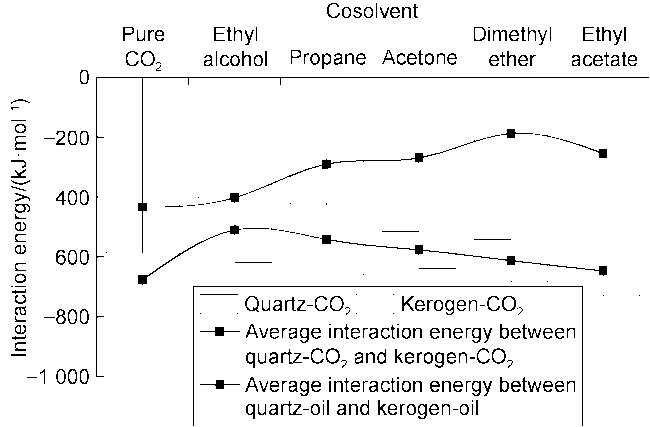
Fig. 7 shows the displacement efficiency of three types of shale oil under the influence of different cosolvents in 10 nm composite pore. The following results can be obtained from this figure. (1) The displacement efficiency of shale oil by pure CO
2 is the lowest, 8.42% for Type I, 13.24% for Type II and 17.23% for Type III. (2) The displacement efficiencies were all significantly enhanced with the addition of cosolvents. Dimethyl ether brought the greatest enhancement, followed by ethyl acetate, and ethanol the worst. This is mainly caused by the strong interaction between ether group and CO
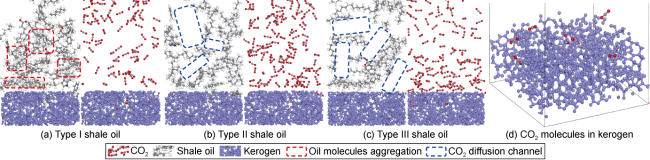
2, and the presence of two methyl groups makes dimethyl ether lipophilic, thus CO
2 is constantly dissolved in shale oil
[18]. CO
2 can be attracted by the carbon-oxygen double bond in the ester group, therefore the solubilization effect of CO
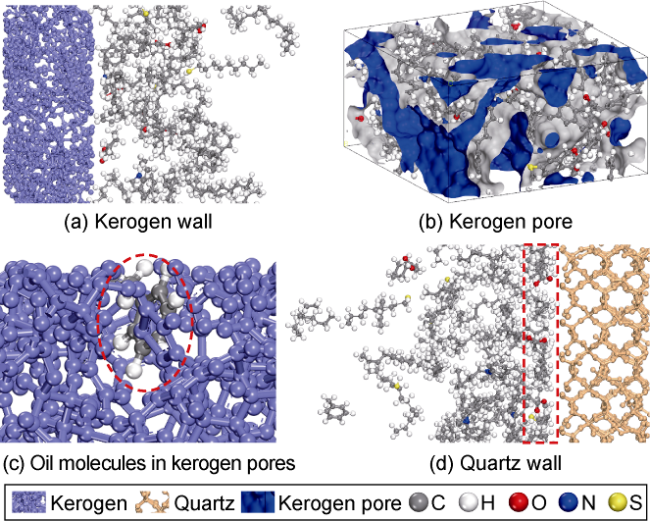
2 is excellent. (3) The benzene ring is strongly attracted by kerogen, and there is also an aromatic ring stacking effect between hydrocarbon molecules with benzene ring structure, which increases the difficulty of stripping shale oil containing benzene rings. (4) Hydrogen bonds are formed between kerogen and cyclohexanepropionic acid or various non-hydrocarbon compounds, which makes it difficult for non-hydrocarbon compounds to be stripped. (5) The effect of CO
2+ethanol displacing Type I shale oil is stronger than that of CO
2+propane, and the effect of CO
2+ethyl acetate displacing Type I shale oil is stronger than that of CO
2+dimethyl ether. However, the effect of CO
2+ethanol and CO
2+ethyl acetate displacing types II and III shale oil is relatively weak. This is because the polarity of hydroxyl and ester groups is stronger than that of alkyl and ether groups, respectively, and with good affinity with non-hydrocarbon compounds. (6) The tripping effect of CO
2+ethyl acetate on Type I shale oil is the best, with a displacement efficiency of 28.26%. By contrast, the stripping effect of CO
2+dimethyl ether on types II and III shale oil is the best, with displacement efficiencies of 41.65% and 43.01%, respectively. (7) The stripping effect of CO
2+ethyl acetate on Type I shale oil is the best, and types II and III shale oil rank second. According to simulations, when temperature increased to 348.15 K and 373.15 K, the displacement efficiency of CO
2+ethyl acetate on Type III shale oil reached 43.81% and 47.63%, respectively; when pressure increased to 30 MPa and 40 MPa, the displacement efficiency on Type III shale oil reached 48.12% and 52.80%, respectively. This is because the increase in temperature results in more intense molecular movement, and the increase in pressure results in an increase in fluid density and a shortening of mass transfer distance between the two phases, which increases the solubility of CO
2+ethyl acetate mixture in shale oil
[18]. Besides, ethyl acetate can reduce the MMP between CO
2 and shale oil and improve their mobility ratio
[7]. Therefore, ethyl acetate is more adaptable as a cosolvent for CO
2.