Introduction
1. Experiment
1.1. Sample
1.2. Method
1.2.1. Evaluation of static adsorption of fracturing fluid thickener
1.2.2. Evaluation of dynamic permeability damage of fracturing fluid thickener
1.2.3. Microscopic characterization of fracturing fluid thickener adsorption morphology
Table 1. Design of AFM adsorption experiment parameters |
| No. | Type | Mass fraction/ % | pH | Temperature/ °C | Remarks |
|---|---|---|---|---|---|
| 1 | Hydroxypropyl guar gum | 0.4 | 9.0 | 20 | Control group |
| 2 | 0.2 | 9.0 | 20 | Mass fraction comparison | |
| 3 | 0.4 | 7.0 | 20 | pH comparison | |
| 4 | 0.4 | 9.0 | 80 | Temperature comparison | |
| 5 | Polyacrylamide | 0.4 | 7.0 | 20 | Control group |
| 6 | 0.2 | 7.0 | 20 | Mass fraction comparison | |
| 7 | 0.4 | 9.0 | 20 | pH comparison | |
| 8 | 0.4 | 7.0 | 80 | Temperature comparison |
1.2.4. Evaluation of electrostatic adsorption of fracturing fluid thickener
1.2.5. Evaluation of adsorption of functional group of fracturing fluid thickener
1.2.6. Evaluation of adsorption of intermolecular force of fracturing fluid thickener
2. Results and discussion
2.1. Static adsorption capacity of fracturing fluid thickener
2.2. Dynamic permeability damage of fracturing fluid thickener
Table 2. Dynamic permeability damage of deep coal rock by fracturing fluid thickener adsorption |
| Type | No. | Porosity/ % | Initial permeability/ 10−3 μm2 | Permeability after adsorption/10−3μm2 | Damage rate/% |
|---|---|---|---|---|---|
| Hydroxy- propyl guar gum | DC-1 | 2.37 | 2.39 | 1.54 | 35.62 |
| DC-2 | 2.32 | 4.13 | 2.60 | 37.01 | |
| DC-5 | 2.60 | 0.45 | 0.29 | 35.24 | |
| Polyacry- lamide | DC-3 | 2.40 | 6.98 | 5.45 | 21.93 |
| DC-4 | 2.34 | 6.00 | 5.14 | 14.30 | |
| DC-6 | 1.39 | 0.39 | 0.32 | 19.63 |
2.3. Microscopic morphology of adsorption layer of fracturing fluid thickener
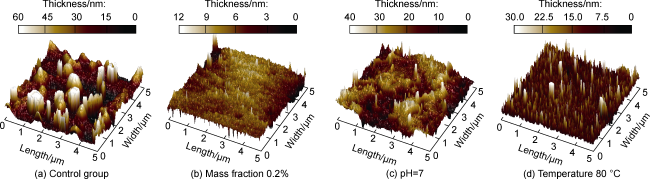
Fig. 1. Three-dimensional morphology of hydroxypropyl guar gum adsorption layer on the surface of deep coal rock. |
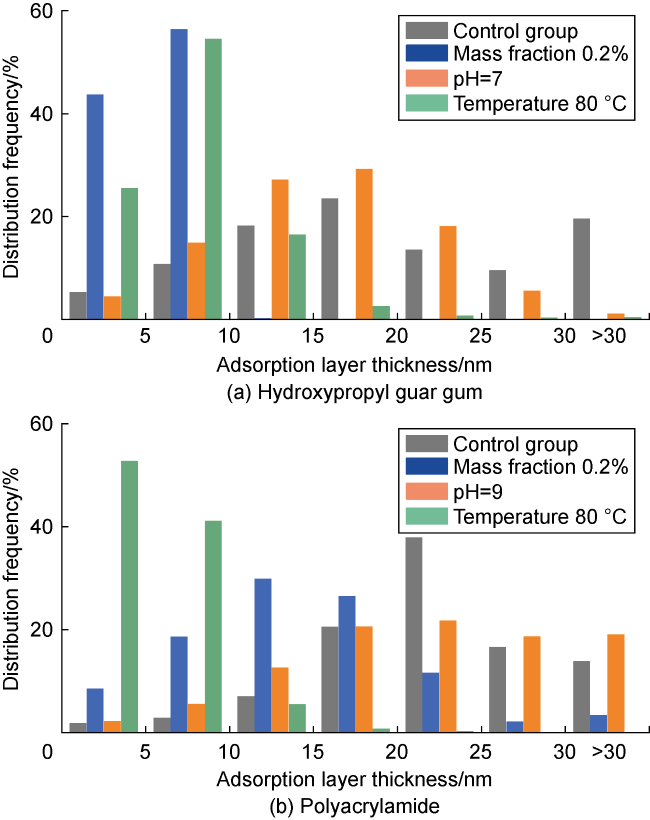
Fig. 2. Thickness distribution of thickener adsorption layer on the surface of deep coal rock. |
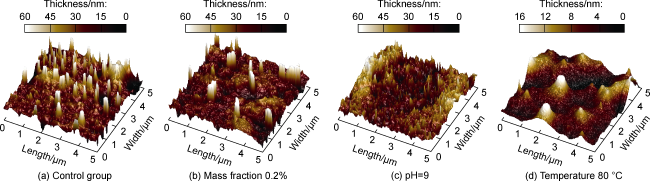
Fig. 3. Three-dimensional morphology of polyacrylamide adsorption layer on the surface of deep coal rock. |
2.4. Electrostatic adsorption of fracturing fluid thickener
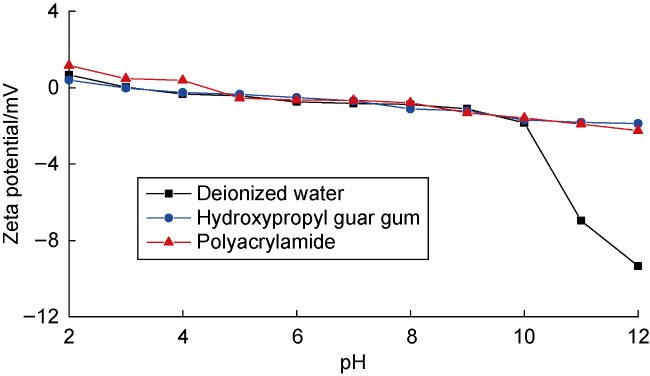
Fig. 4. Zeta potential of deep coal rock versus pH value in different fluids. |
2.5. Functional group adsorption of fracturing fluid thickener
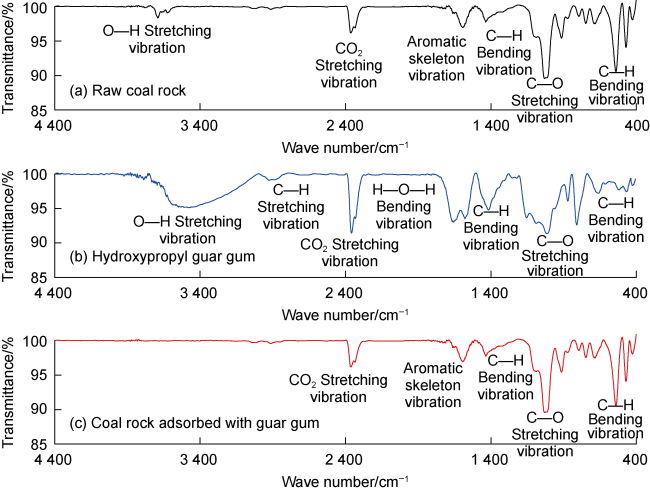
Fig. 5. Infrared spectral curve of coal rock adsorbed with hydroxypropyl guar gum. |
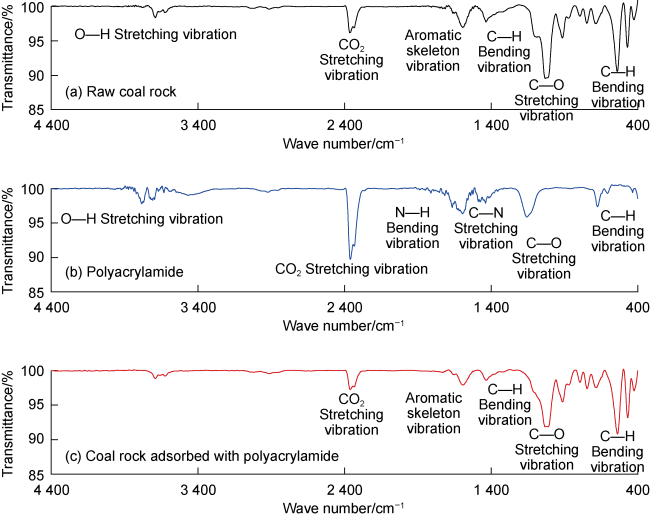
Fig. 6. Infrared spectral curve of coal rock adsorbed with polyacrylamide. |
2.6. Intermolecular forces of fracturing fluid thickener
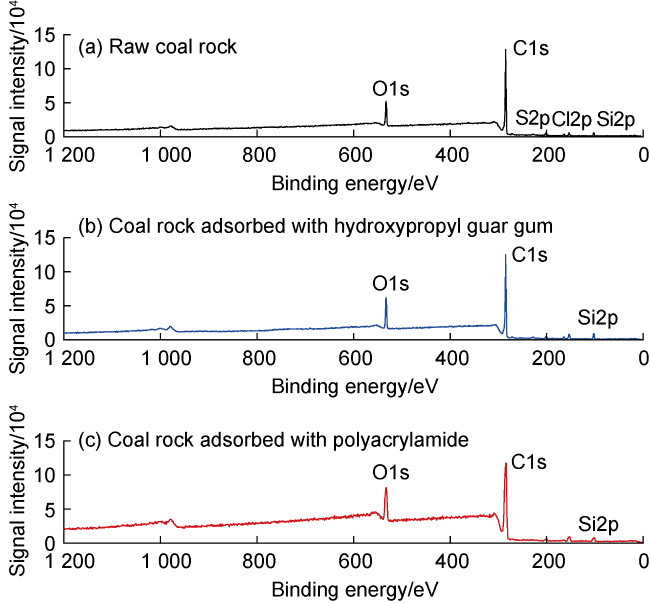
Fig. 7. Full-wave band XPS analysis of coal rock. O1s -electrons in 1s orbital of oxygen atoms; C1s -electrons in 1s orbital of carbon atoms; S2p -electrons in 2p orbital of sulfur atoms; Cl2p -electrons in 2p orbital of chlorine atoms; Si2p -electrons in 2p orbital of silicon atoms. |
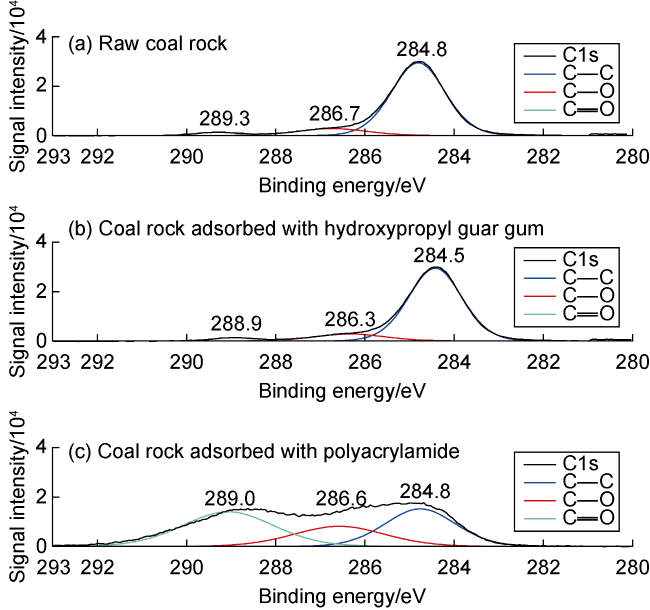
Fig. 8. Peak fitting in C1s band of XPS high-resolution spectra. |
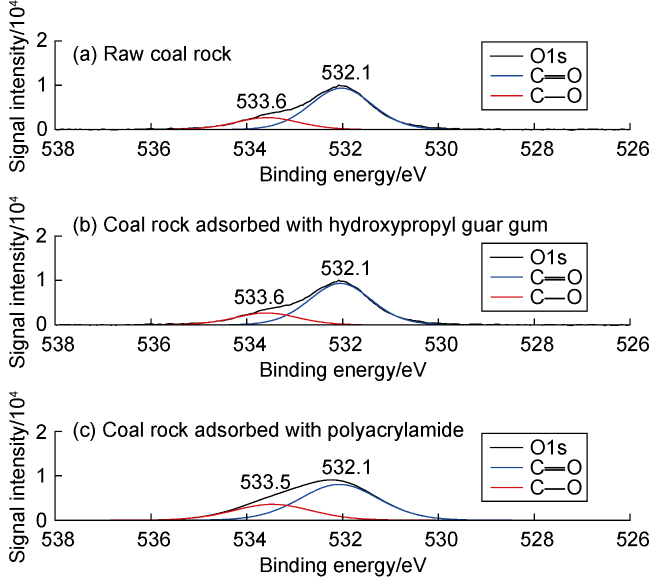
Fig. 9. Peak fitting in O1s band of XPS high-resolution spectra. |
3. Adsorption damage mechanism and control countermeasures of fracturing fluid thickener in deep coal rock
3.1. Adsorption mechanism of fracturing fluid thickener in deep coal rock
3.1.1. Electrostatic adsorption in deep coal rock
3.1.2. Functional groups adsorption in deep coal rock
3.1.3. Intermolecular forces between coal rock and fracturing fluid thickener
3.2. Adsorption damage mechanism of fracturing fluid in deep coal rock
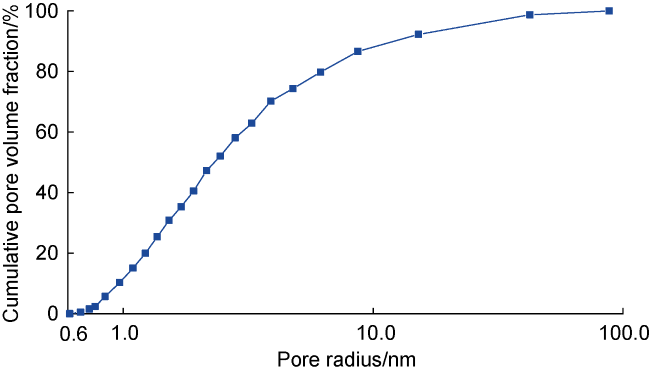
Fig. 10. Pore radius distribution of nanopores in deep coal rocks measured by nitrogen adsorption experiment. |
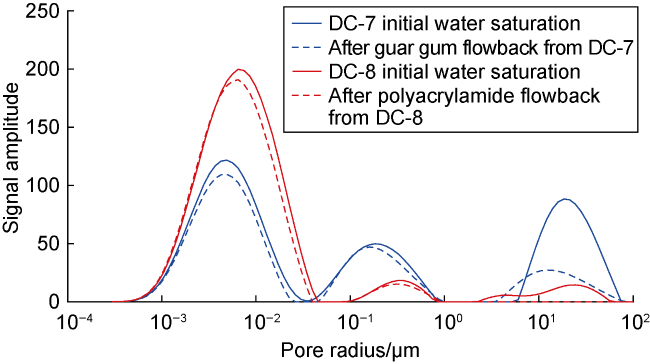
Fig. 11. Nuclear magnetic resonance T2 spectra of deep coal rock before and after fracturing fluid flowback by gas displacement. |
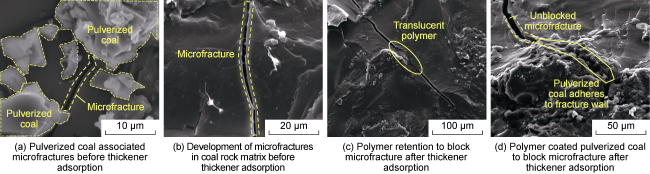
Fig. 12. Scanning electron microscope morphology of microfractures before and after fracturing fluid intrusion into deep coal rock. |