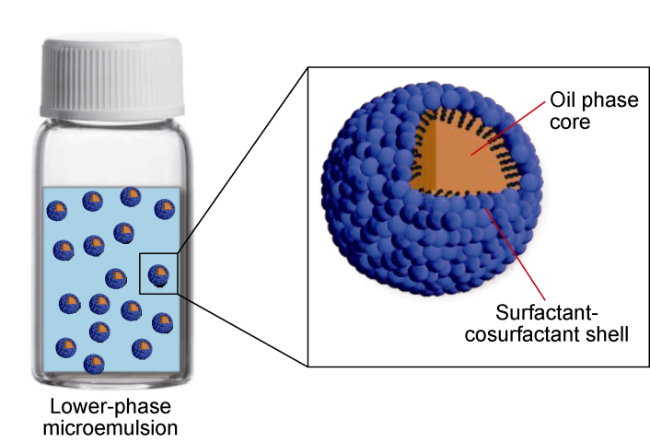
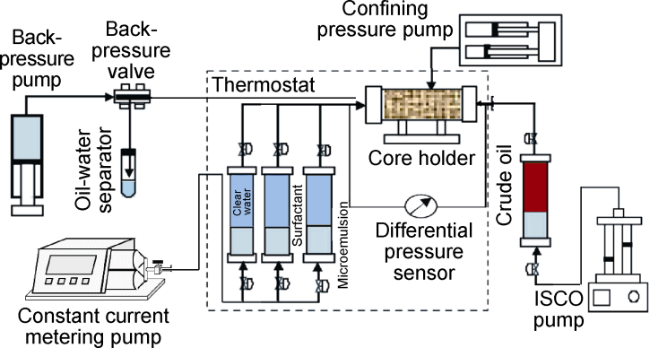
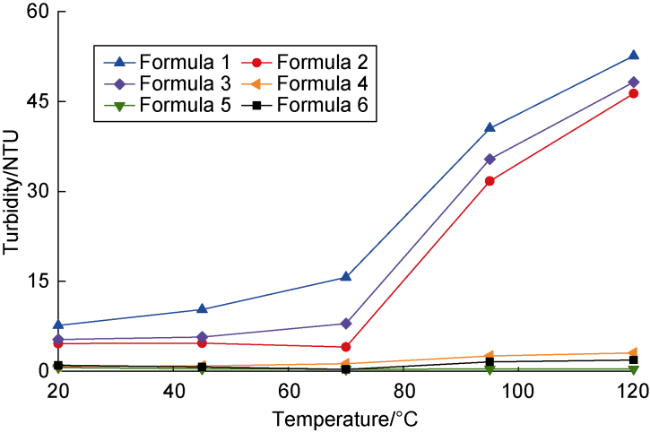
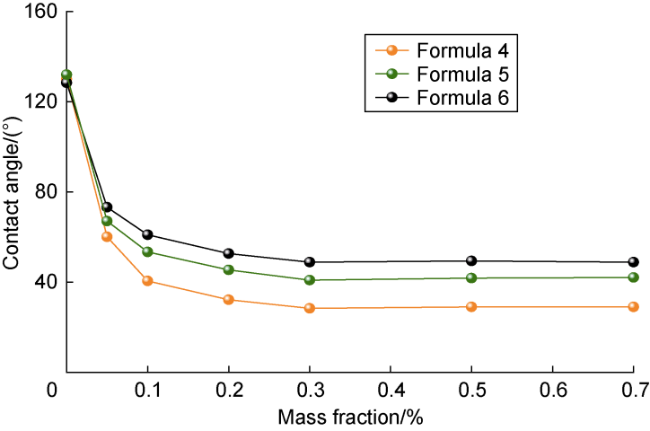
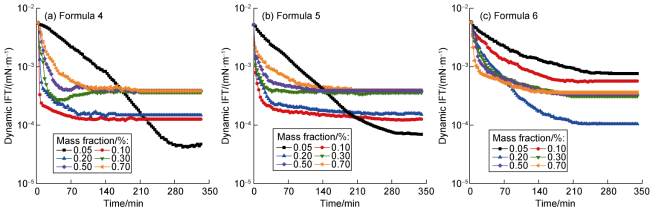
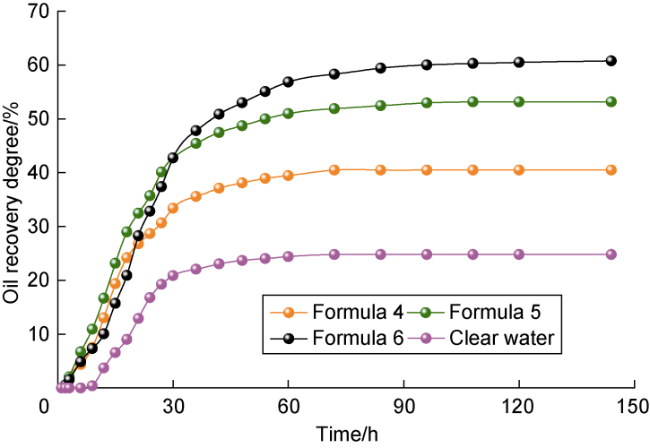
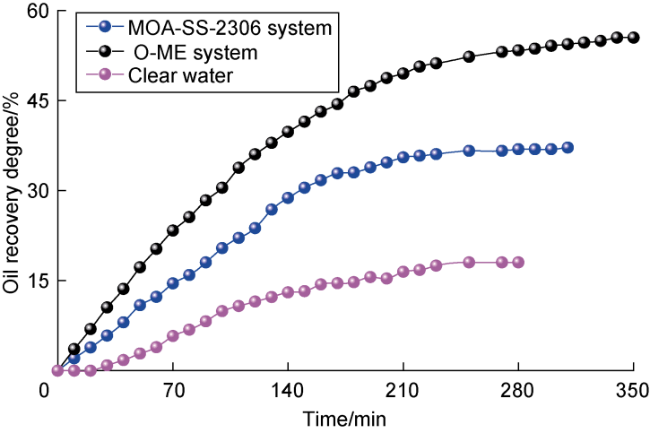
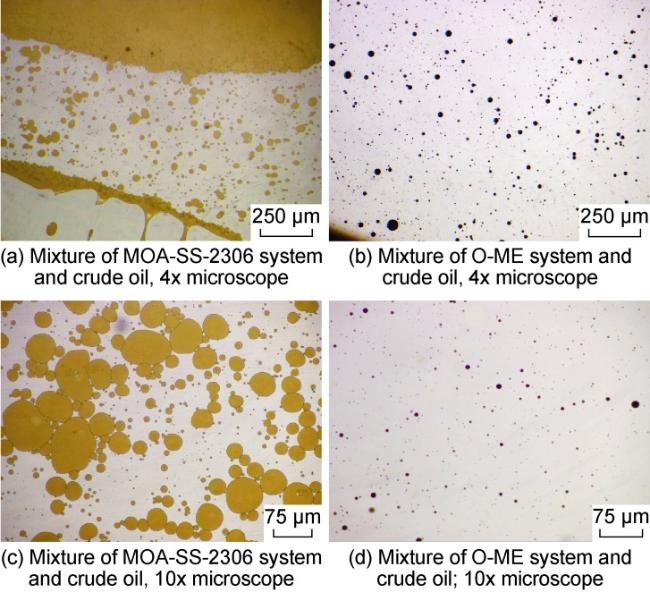
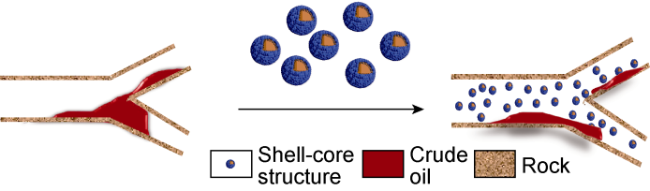
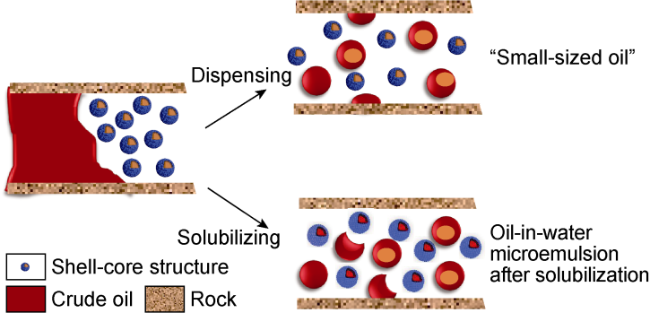
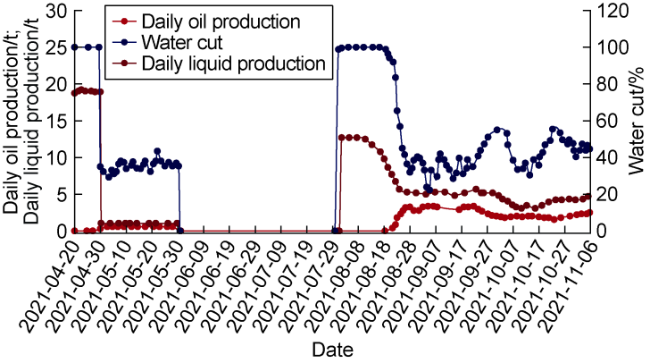
Ultra-low permeability and tight reservoirs have low porosity and permeability, as well as complex pore throat structures. Fluid displacement in formations mainly depends on imbibition, that is, the wetting phase fluid displaces the non-wetting phase fluid under the combined action of interfacial tension (IFT), gravity, and capillary pressure differences between pore channels
[1-2]. For water-wet ultra-low permeability and tight reservoirs, the imbibition agent enters the pore throat under the action of capillary pressure, replacing and recovering crude oil by spontaneous imbibition. Therefore, spontaneous imbibition can significantly enhance oil recovery (EOR) and become an important mechanism of EOR in ultra-low permeability and tight reservoirs
[3]. However, the wettability of most reservoirs tends to be oleophilic due to the existence of acidic substances in crude oil. As a resistance, the capillary pressure greatly inhibits the imbibi-tion oil displacement effect
[4]. In order to improve the oil recovery of oil-wet ultra-low permeability and tight reservoirs, a large number of experimental studies on enhancing imbibition oil recovery have been carried out by scholars at home and abroad, including CO
2 huff and puff, surfactant huff and puff, and nanofluid displacement
[5]. Because of its low IFT and good wettability alteration ability, surfactant solution has a great application potential in enhancing spontaneous imbibition oil recovery (SIOR) in ultra-low permeability and tight reservoirs. However, there is a more serious loss in adsorption, retention and precipitation of surfactants during the process of migration, which greatly reduces the migration distance and oil displacement efficiency. Therefore, the development of spontaneous imbibition agents with low adsorption loss, high spontaneous imbibition oil recovery, and suitable for ultra-low permeability and tight reservoirs is of great significance to improve the oil recovery of ultra-low permeability and tight reservoirs
[6-7].