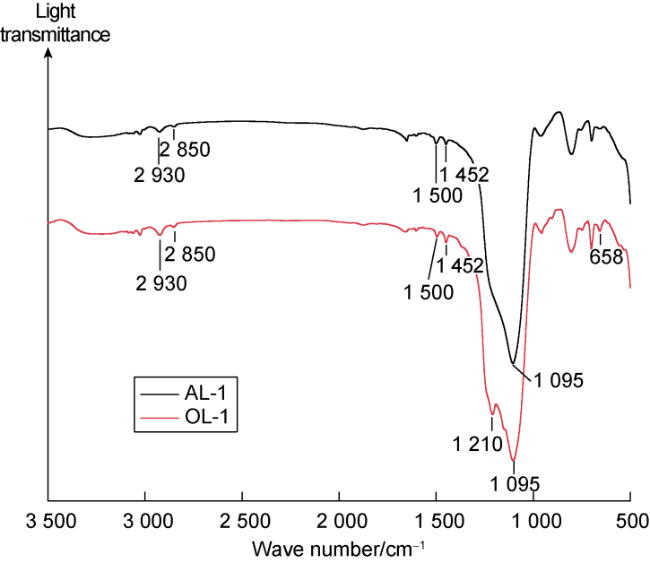
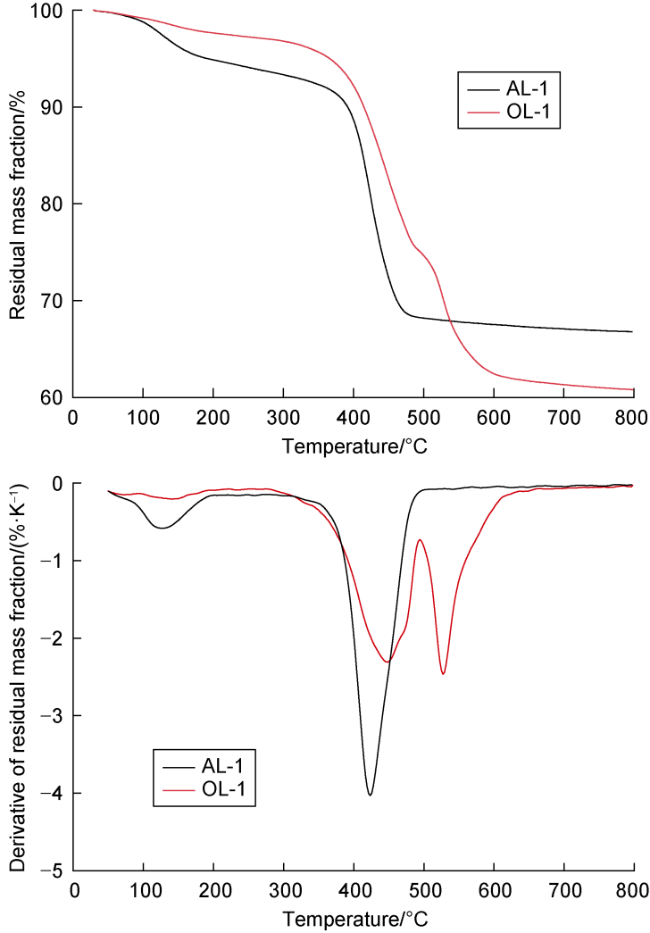
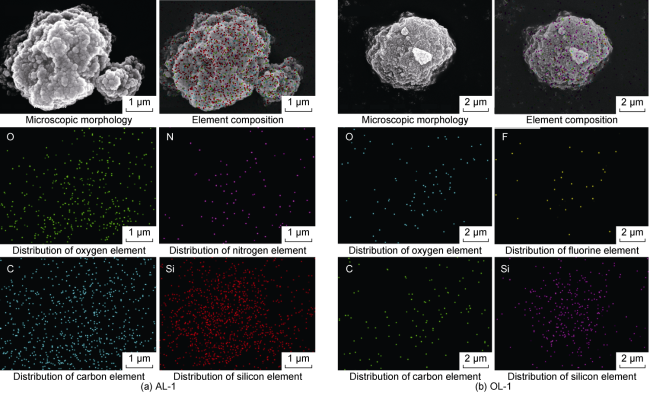
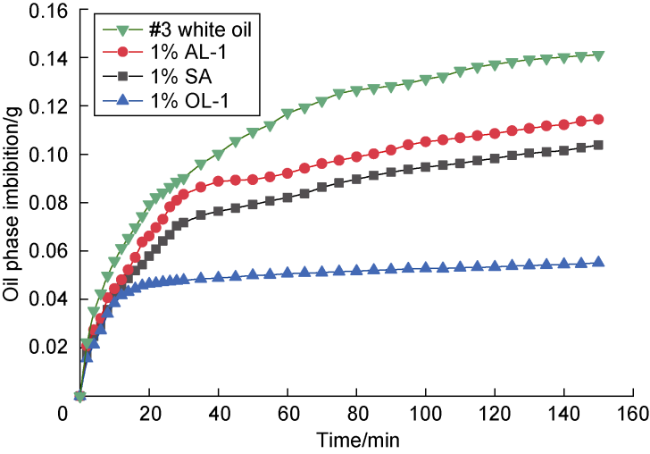
Many submicron fractures and pores are developed in shale. Meanwhile, its lipophilicity is relatively strong
[8], inducing a high capillary pressure to drive the oil phase to invade the shale. When the drilling fluids column pressure is significantly higher than the formation pore pressure, the oil phase intrusion would be more serious. Oil phase intrusion, on the one hand, triggers the transfer of drilling fluids column pressure in the formation, which can increase the formation pore pressure and easily cause the formation to crack or spall along the bedding planes or weak interfaces
[9]. On the other hand, organic matters such as kerogen and asphalt in formation are prone to swell or even be dissolved after contacting oil phase, causing uneven stress distribution in the rock and further aggravating wellbore instability
[7]. The intrusion of oil phase from OBDF into formation is the crucial cause for wellbore instability caused by drilling fluids
[10]. Many researches have been carried out for many years at home and abroad on wellbore instability caused by oil phase intrusion, which is mainly focused on plugging technology to inhibit oil phase intrusion and thus improve wellbore stability. The existing plugging agents mainly include conventional micro- and nano-sized agents. However, it is hard to plug the micro- and nano-pores/ fractures in shale using conventional micro-sized plugging agents
[11]. In contrast, due to the large specific surface area and high surface energy, the nano-sized plugging agents aggregate easily after being added to the non-polar oil phase, and largely increase in size, which may weaken the plugging effect on the micro- and nano-pores in shale
[12-13]. Therefore, present plugging technology fails to eliminate wellbore instabilities caused by oil phase intrusion.