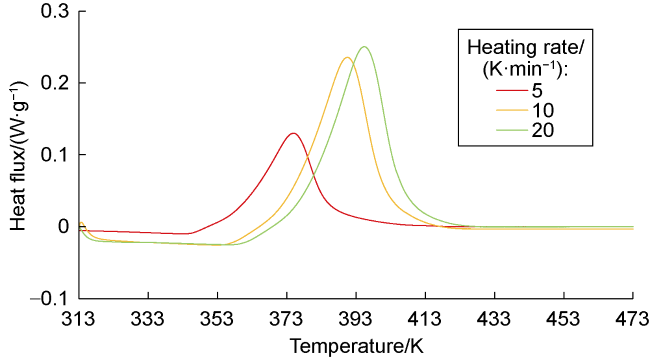
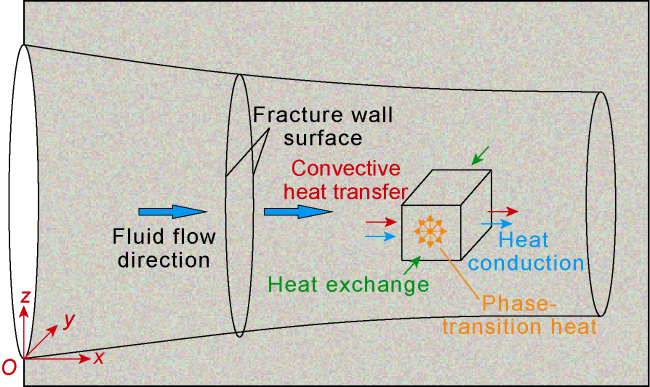
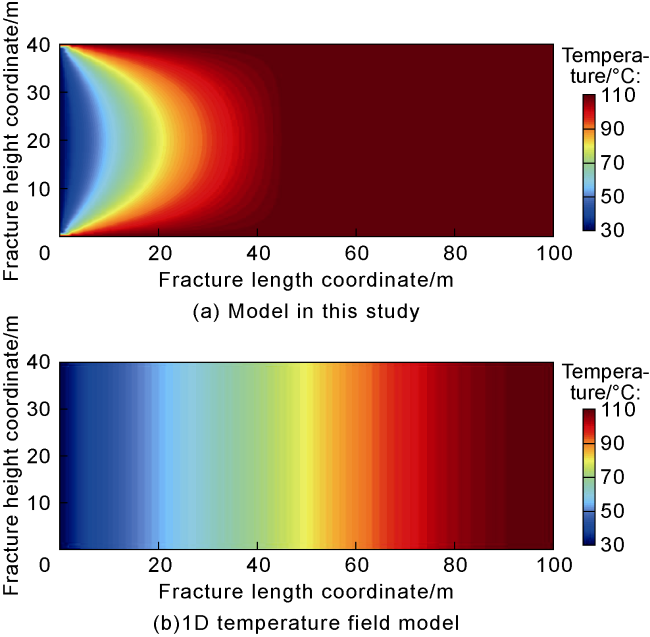
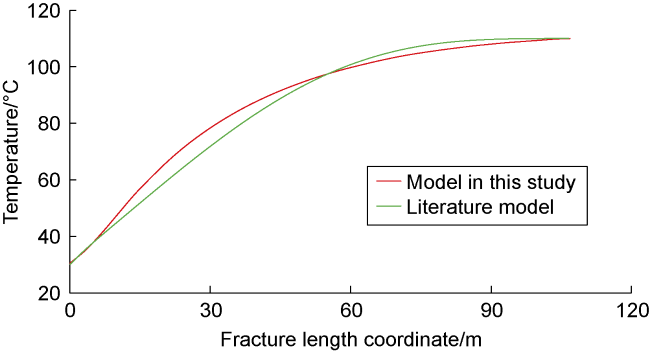
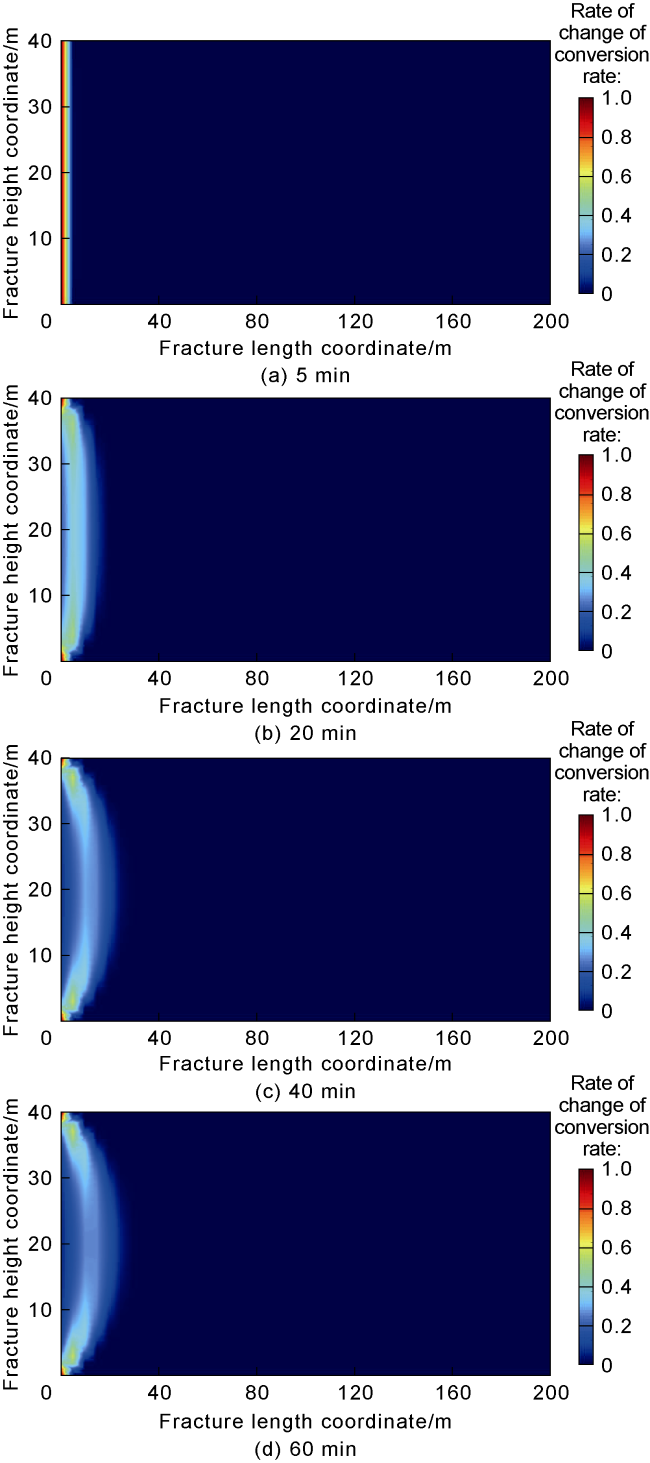
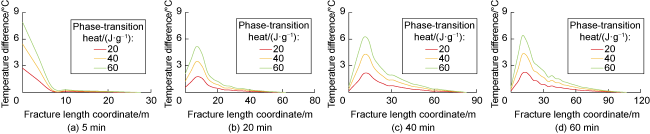
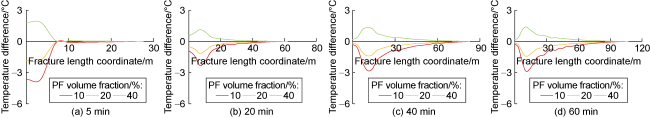
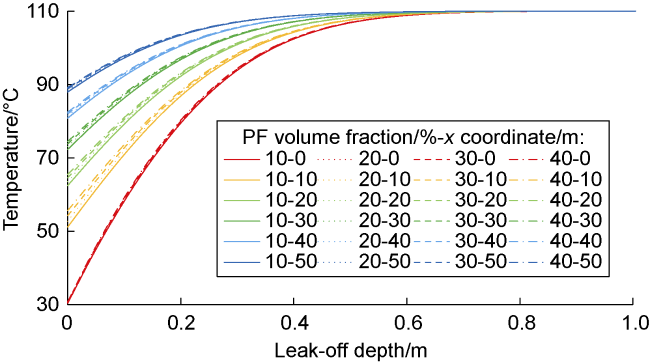
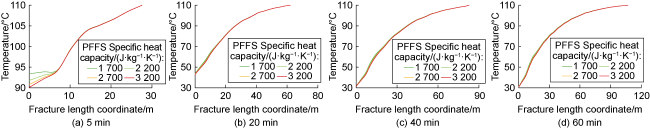
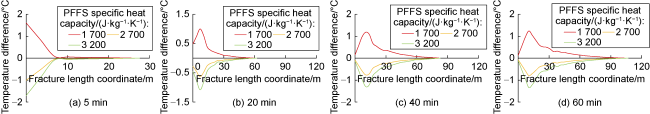
Phase-transition heat refers to the amount of heat released by PF per unit mass (
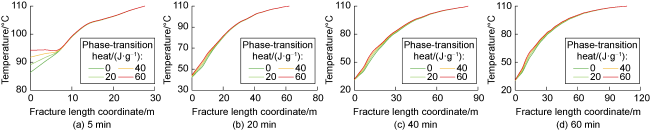
Table 1). The phase-transition heat values measured under heating rates of 5, 10, and 20 K/min are 32.07, 31.40, and 32.10 J/g, respectively, with an average value of 31.86 J/g. To analyze the influence of phase-transition heat on the fracture temperature field, the temperature distributions under phase-transition heat values of 0, 20, 40, and 60 J/g were calculated (
Fig. 7). The results indicate that the larger the phase-transition heat, the higher the relative temperature within the fracture. Furthermore, the temperature difference near the fracture mouth is significant under different phase- transition heat conditions, while the temperature difference decreases as the distance to the fracture tip increases. At injection times of 5, 20, 40, and 60 min, the maximum temperature difference occurs at approximately 0, 7, 10, and 10 m from the fracture mouth along the central axis under different phase-transition heat conditions. This phenomenon can be attributed to the following reasons: (1) At an injection time of 5 minutes, the overall temperature within the fracture is relatively high. PF undergoes substantial phase transition near the fracture mouth, and the resulting phase-transition heat significantly raises the temperature at the mouth (
Fig. 7a). (2) At an injection time of 20 min, the temperature at the fracture mouth is lower, and the temperature gradually increases along the fracture length. The temperature at the mouth is below the critical phase-transition temperature of PF, resulting in a lower phase transition rate and limited temperature increase at the mouth. However, at approximately 7 m within the fracture, the temperature rises to the critical value for substantial PF phase transition, causing a significant temperature increase due to the release of phase-transition heat. Beyond 7 m, the remaining PF and phase-transition volume decrease, leading to a reduced temperature variation caused by phase-transition heat (
Fig. 7b). Sustained injection of low- temperature fracturing fluid does not cause an indefinite temperature drop but rather leads to a gradual stabilization
[17]. (3) At injection times of 40 and 60 min, the temperature differences within the fracture under different phase-transition heat conditions are minimal. The temperature at the fracture mouth remains low, with limited PF undergoing phase transition, resulting in a small temperature rise. At approximately 10 m within the fracture, the temperature rises sufficiently to trigger substantial PF phase transition. Beyond 10 m, the remaining PF and phase-transition volume are minimal, reducing the temperature variation caused by phase-transition heat (
Fig. 7c and 7d).