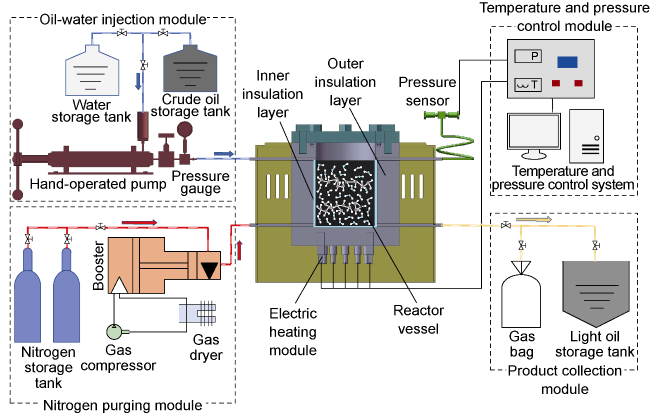
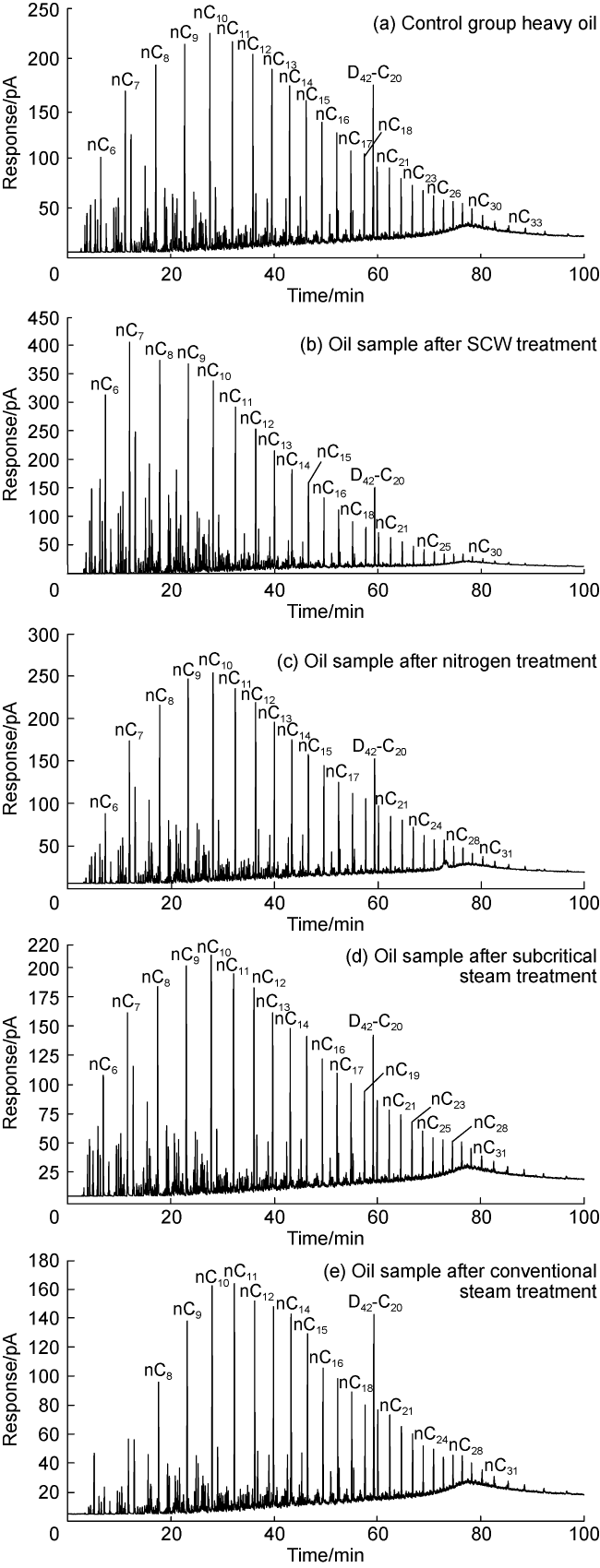
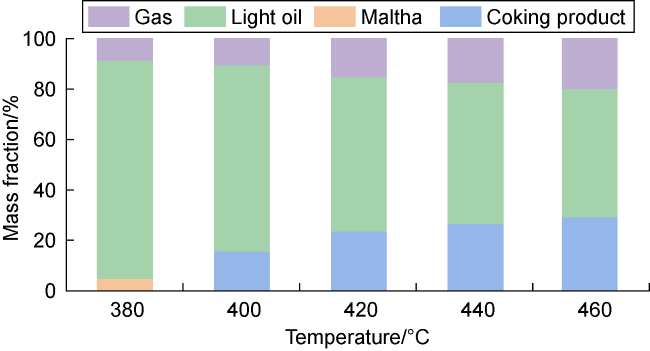
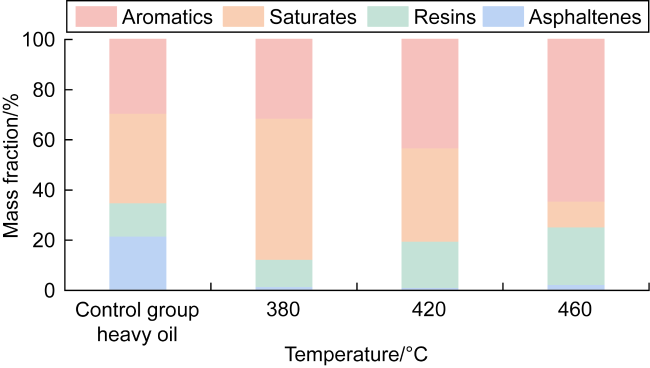
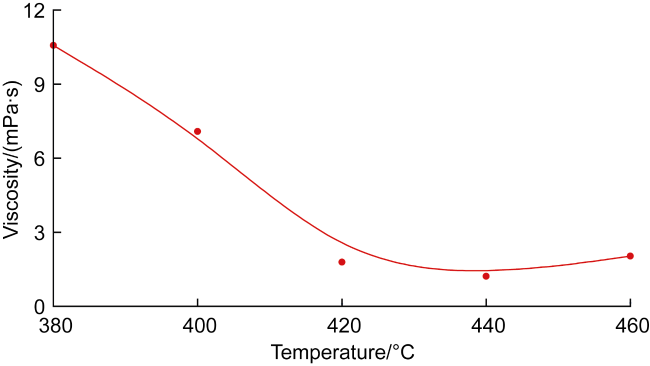
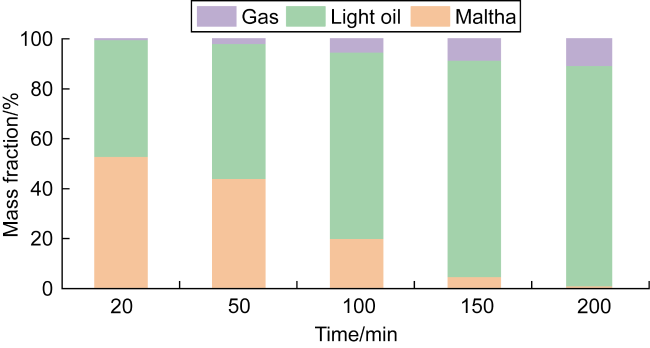
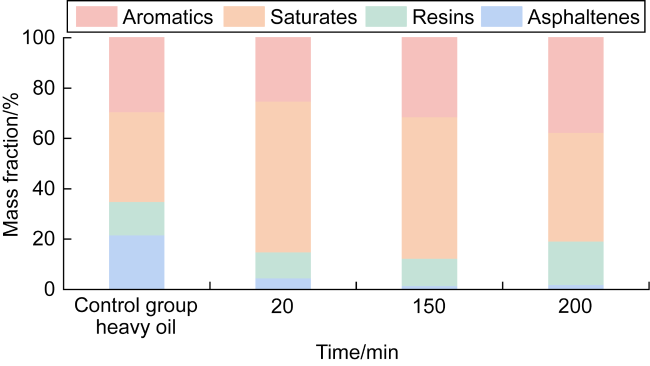
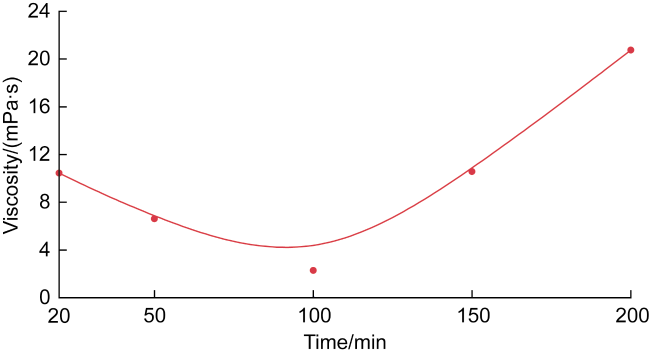
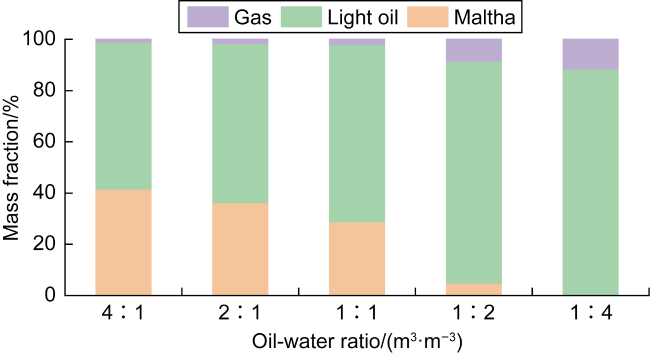
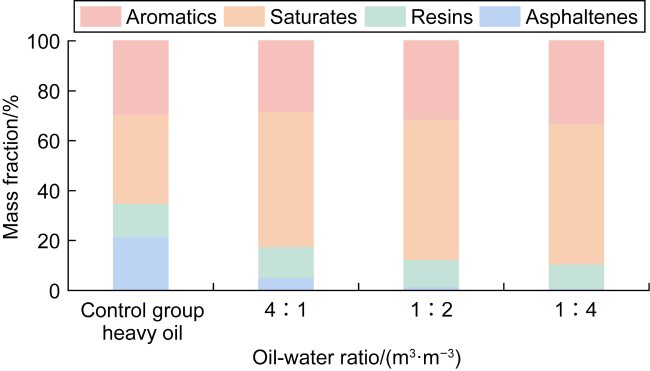
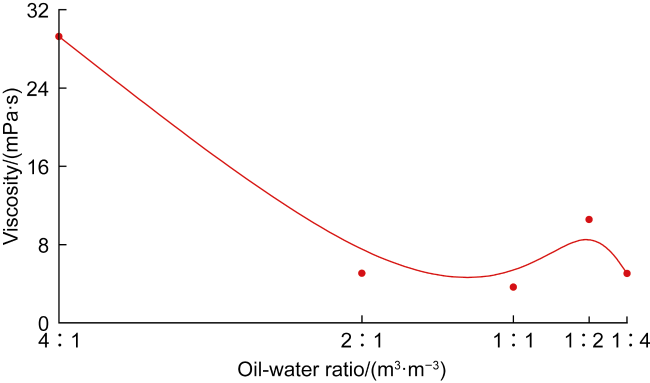
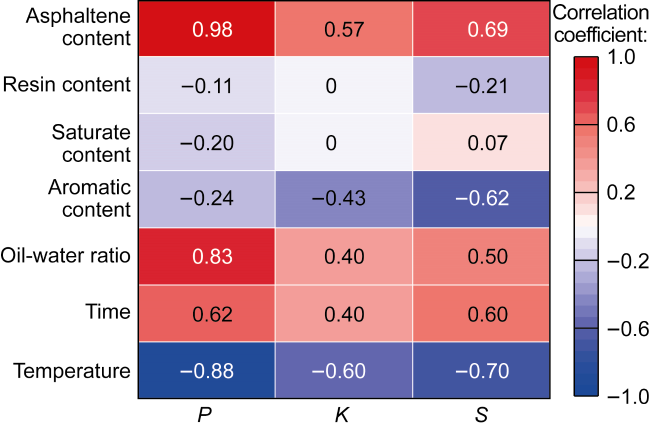
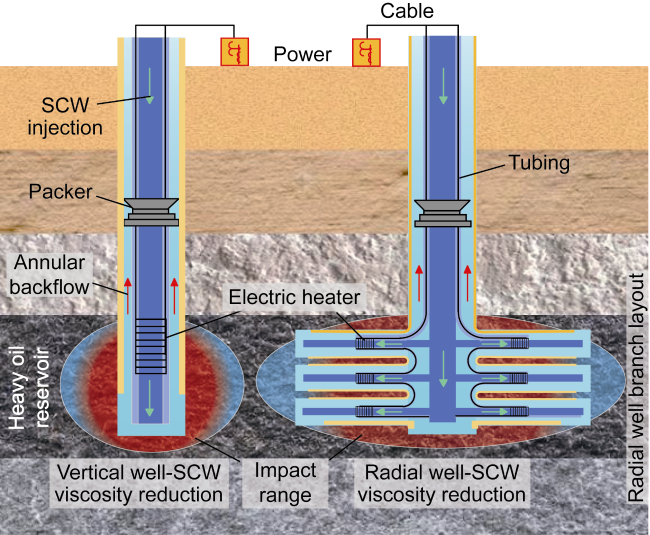
In recent years, the method of upgrading heavy oil using supercritical water (SCW) has garnered widespread attention. SCW refers to water at temperatures exceeding 374 °C and pressures above 22.1 MPa
[3]. It is characterized by low viscosity, strong diffusivity, and high density
[4⇓-6], which can accelerate the reaction rate of heavy oil upgrading and shorten reaction times
[7-8], while significantly reducing coke formation rates
[9]. The use of SCW for viscosity reduction offers the advantage of physically separating crude oil products without relying on chemical additives or catalysts. After sedimentation and dewatering, the treated crude oil can be transported without requiring heating stations along the pipeline, and reducing transportation costs. Zhao et al. discovered that supercritical multi-component thermal fluids (organic wastewater mixed with gasified supercritical water) effectively promote the in-situ transformation of underground heavy oil and low-maturity shale oil, significantly enhancing the hydrocarbon conversion efficiency of organic matter
[10]. Dong et al. conducted kinetic studies on SCW-based heavy oil upgrading, identifying three reaction stages: the induction phase, cracking phase, and gas generation phase, established a six-lump kinetic model incorporating saturates, aromatics, resins, asphaltenes, coke, and gases
[11]. Liu investigated the effect of SCW on the thermal cracking of asphaltenes, revealing that asphaltenes can form cavity-like structures in SCW and self-assemble into clusters, significantly accelerating the condensation and coke formation reactions
[12]. In summary, previous studies on the upgrading of heavy oil using SCW have primarily focused on reaction kinetics and coking behavior, with limited systematic evaluation of the characteristics of the produced fluids and viscosity reduction. The key factors influencing the effectiveness of SCW upgrading and viscosity reduction, as well as the extent of their impact, remain unclear. Further research is needed to elucidate the microscopic mechanisms and dominant controlling factors.