Introduction
1. Basic methods for formation water identification and data credibility screening
1.1. Physical characteristic analysis
1.2. Chemical composition analysis
1.3. Sulin water type analysis
1.4. Single hydrochemical characteristic coefficient analysis
1.4.1. Sodium chloride coefficient analysis
Table 1. Formation closed environment discrimination and hydrochemical data credibility evaluation by sodium chloride coefficient |
| Water type | Sodium chloride coefficient | Description | ||
|---|---|---|---|---|
| Closed environment | Relatively closed environment | Open environment | ||
| CaCl2-type water | <0.65 | 0.65-0.85 | 0.85-1.00 | When the geological setting analysis confirms that the geological environment is consistent with it, the data are credible; otherwise, the data are incredible. In addition, when influenced by condensate water or ancient atmospheric fresh water, the sodium chloride coefficient can be greater than 0.85. |
| NaHCO3-type water (onshore petroliferous basins) | 1.00-2.00 | 2.00-5.00 | >5.00 | When the geological setting analysis confirms that the geological environment is consistent with it, the data are credible; otherwise, the data are incredible. In addition, the sodium chloride coefficient of the water in the formation that was closed again after the opening in the Himalayan period can reach 5-10. |
1.4.2. Desulfurization coefficient analysis
Table 2. Formation closed environment discrimination and hydrochemical data credibility evaluation by desulfurization coefficient |
| Water type | Desulfurization coefficient | Description | ||
|---|---|---|---|---|
| Closed environment | Relatively closed environment | Open environment | ||
| CaCl2-type water | <1 | 1-3 | >3 | When the geological setting analysis confirms that the geological environment is consistent with it, the data are credible; otherwise, the data are incredible. In addition, when anaerobic bacterial desulfurization is complete near the redox interface, or when there is a large number of Ba2+ in the formation water, the desulfurization coefficient can be zero. |
| NaHCO3-type water | <20 | 20-40 | >40 | |
| Surface seawater | <1 | 1-10 | >10 | |
2. Comprehensive evaluation method and influencing factors for the data credibility of formation water
2.1. Comprehensive evaluation on the sampling process
2.2. Comprehensive evaluation and influencing factors of the hydrochemical data credibility
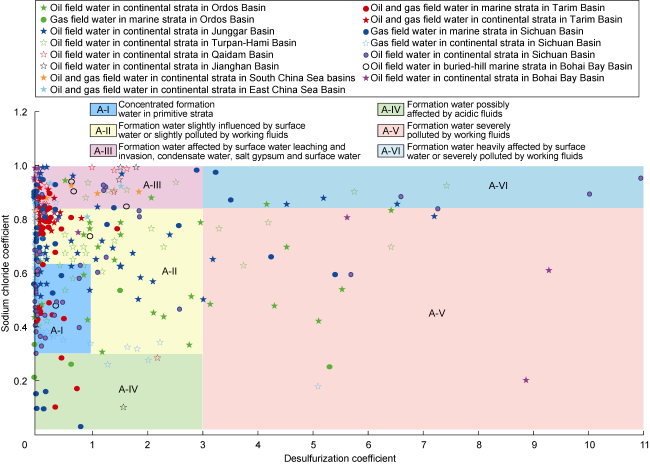
2.2.1. CaCl2-type water
Fig. 1. Classification and influencing factors of CaCl2-type formation water in oil and gas fields of petroliferous basins in China. |
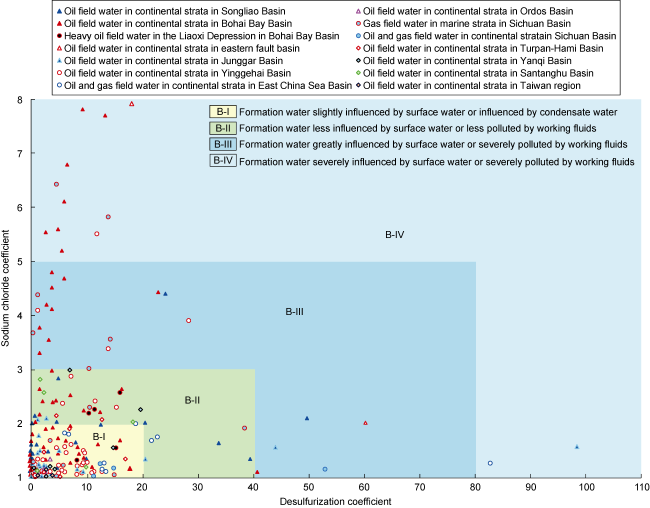
2.2.2. NaHCO3-type water
Fig. 2. Classification and influencing factors of NaHCO3-type formation water in oil and gas fields of petroliferous basins in China. |