Introduction
1. Experimental samples and methods
2. Results
2.1. Total organic carbon, maturity, and dispersed organic matter type
Table 1. Organic petrographic characteristics of overmature shale samples from the Longmaxi Formation, Sichuan Basin |
| Well | Depth/ m | Sample ID | TOC/ % | BRo/ % | GRo/ % |
|---|---|---|---|---|---|
| W1 | 3 742.00 | W1-1 | 1.83 | 2.42 | 2.68 |
| W1 | 3 743.26 | W1-2 | 5.03 | 2.45 | 2.62 |
| Y1 | 4 145.55 | Y1 | 4.31 | 3.42 | 3.22 |
| N1 | 3 591.84 | N1 | 3.16 | 3.69 | 3.69 |
Fig. 1. Characteristics of graptolite periderms and pyrobitumen under oil-immersion microscopy in the Longmaxi Formation shales, Sichuan Basin. (a) Well W1, Sample W1-1, 3 742.00 m; (b) Well Y1, Sample Y1, 4 145.55 m. |
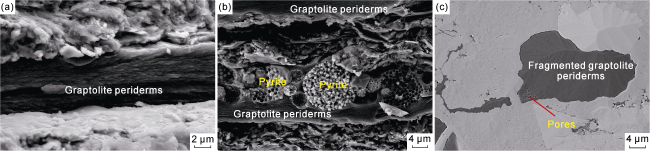
Fig. 2. SEM images of graptolite periderms in Longmaxi shales, Sichuan Basin (Well W1, Sample W1-1, 3 742.00 m). (a) Graptolite periderms distributed along bedding planes, fresh rock cross-section; (b) Framboidal pyrite infilling within non-granular graptolite periderms, fresh rock cross-section; (c) Fragmented graptolite periderms with local pores, polished surface. |
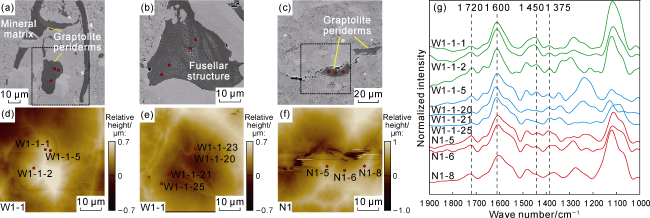
Fig. 3. SEM images, surface morphology, and AFM-IR spectra of graptolite periderms in the Longmaxi Formation shales, Sichuan Basin (red dots indicate AFM-IR single-point test locations). (a) SEM image of fragmented graptolite periderms, Sample W1-1, Well W1, 3 742.00 m; (b) SEM image of fragmented graptolite periderms showing fusellar structure, with alternating layers of pore-rich and pore-poor regions, Sample W1-1, Well W1, 3 742.00 m; (c) SEM image of graptolite periderms, Sample N1, Well N1, 3 591.84 m; (d) Morphology of the area marked by the dashed box in |
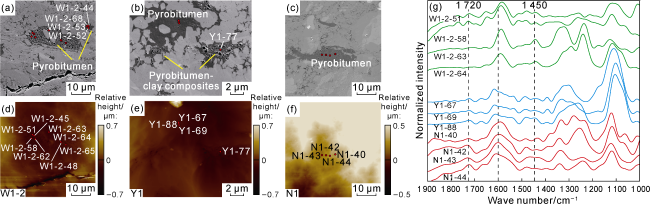
Fig. 4. SEM images, surface morphology, and AFM-IR spectra of homogeneous pyrobitumen in the Longmaxi Formation shales, Sichuan Basin (red dots indicate AFM-IR single-point test locations). (a) Extremely porous region, Sample W1-2, Well W1, 3 743.26 m, SEM; (b) Coexistence of homogeneous pyrobitumen and pyrobitumen-clay composites, with poorly developed pores in homogeneous pyrobitumen, Sample Y1, Well Y1, 4 145.55 m, SEM; (c) Poorly developed pores, Sample N1, Well N1, 3 591.84 m, SEM; (d) Surface morphology of the corresponding area in |
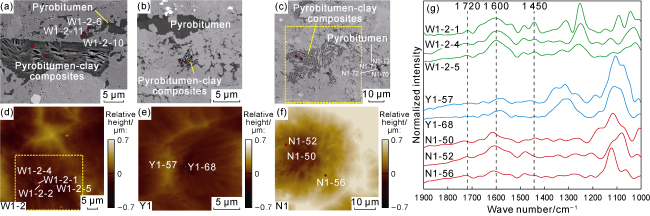
Fig. 5. SEM images, surface morphology, and AFM-IR spectra of pyrobitumen-clay composites in the Longmaxi Formation shales, Sichuan Basin (red dots indicate AFM-IR single-point test locations). (a) Fracture-shaped pores developed along the edges of flake-shaped clay minerals, Sample W1-2, Well W1, 3 743.26 m, SEM; (b) Pyrobitumen-clay composites with highly developed pores, Sample Y1, Well Y1, 4 145.55 m, SEM; (c) Highly porous region, Sample N1, Well N1, 3 591.84 m, SEM; (d) Surface morphology of the sample, Sample W1-2, with the dashed box corresponding to |
2.2. Molecular functional group characterization of dispersed organic matter via AFM-IR spectroscopy
2.3. Organic matter pores
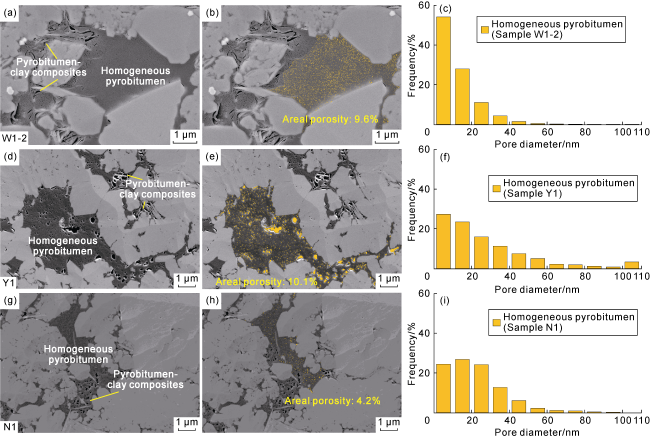
Fig. 6. Pore and pore size development characteristics of typical homogeneous pyrobitumen in the Longmaxi Formation shales, Sichuan Basin (the yellow zones are pores). (a) Homogeneous pyrobitumen and pyrobitumen-clay composites with moderately developed pores, Sample W1-2, Well W1, 3 743.26 m, SEM; (b) Extracted image of pores within homogeneous pyrobitumen particles in |
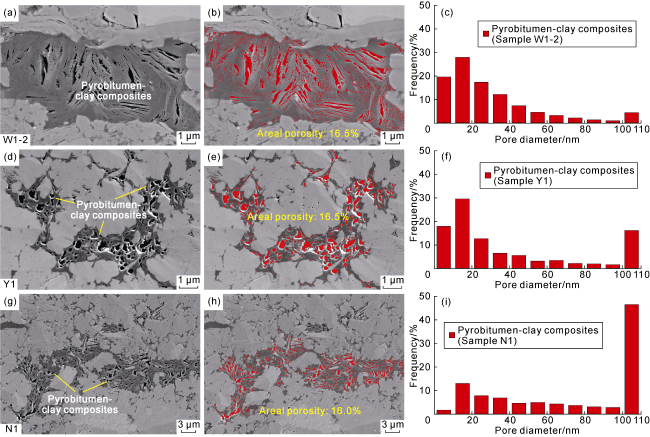
Fig. 7. Pore and pore size development characteristics of typical Pyrobitumen-clay composites in the Longmaxi Formation shales, Sichuan Basin (the red zones are pores). (a) Pyrobitumen-clay composites with fracture-like pores along clay edges, SEM, Sample W1-2, Well W1, 3 743.26 m; (b) Extracted image of pores within pyrobitumen-clay composite particles in |
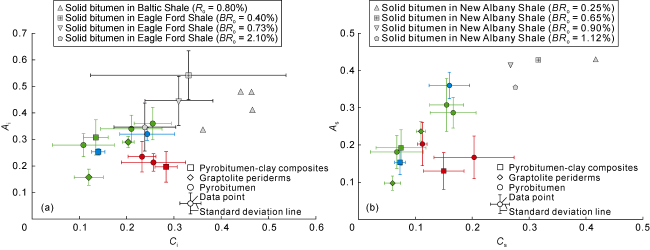
Fig. 8. Distribution characteristics of A-factor and C-factor for different organic matter types in the Longmaxi Shale, Sichuan Basin (Green: Well W1; Blue: Well Y1; Red: Well N1; Solid bitumen data of Baltic and Eagle Ford Shales are from Reference [27]; those of New Albany Shales are from Reference [3]). |
Table 2. AFM-IR spectral parameters and pore development characteristics of typical organic matter in the Longmaxi Formation shales |
| Well | Depth/m | Test point ID | Maceral | Ai-factor | Ci-factor | As-factor | Cs-factor | Pore development characteristics |
|---|---|---|---|---|---|---|---|---|
| W1 | 3 742.00 | W1-1-1 | Graptolite periderms | 0.31 | 0.22 | 0.26 | 0.12 | Fewer pores, |
| W1-1-2 | 0.30 | 0.19 | 0.25 | 0.10 | ||||
| W1-1-5 | 0.26 | 0.20 | 0.20 | 0.11 | ||||
| W1-1-20 | Graptolite periderms | 0.20 | 0.14 | 0.12 | 0.06 | More pores with an areal porosity of 8.5%, | ||
| W1-1-21 | 0.14 | 0.12 | 0.07 | 0.06 | ||||
| W1-1-23 | 0.17 | 0.15 | 0.09 | 0.08 | ||||
| W1-1-25 | 0.12 | 0.07 | 0.11 | 0.04 | ||||
| W1 | 3 743.26 | W1-2-45 | Homogeneous pyrobitumen | 0.34 | 0.18 | 0.23 | 0.12 | Extremely developed pores with lager pore diameter and an areal porosity of 28.2%, |
| W1-2-48 | 0.31 | 0.12 | 0.21 | 0.09 | ||||
| W1-2-51 | 0.25 | 0.20 | 0.19 | 0.14 | ||||
| W1-2-58 | 0.19 | 0.18 | 0.11 | 0.09 | ||||
| W1-2-62 | 0.27 | 0.03 | 0.15 | 0.01 | ||||
| W1-2-63 | 0.30 | 0.05 | 0.15 | 0.02 | ||||
| W1-2-64 | 0.31 | 0.06 | 0.25 | 0.02 | ||||
| W1-2-65 | 0.26 | 0.05 | 0.16 | 0.05 | ||||
| W1-2-44 | Homogeneous pyrobitumen | 0.45 | 0.32 | 0.31 | 0.15 | More pores with small pore diameter and an areal porosity of 9.5%, | ||
| W1-2-52 | 0.37 | 0.25 | 0.40 | 0.19 | ||||
| W1-2-53 | 0.28 | 0.22 | 0.20 | 0.11 | ||||
| W1-2-68 | 0.34 | 0.23 | 0.32 | 0.17 | ||||
| W1-2-6 | Homogeneous pyrobitumen | 0.27 | 0.18 | 0.28 | 0.20 | More pores with small pore diameter and an areal porosity of 9.6%, | ||
| W1-2-10 | 0.36 | 0.30 | 0.34 | 0.19 | ||||
| W1-2-11 | 0.39 | 0.15 | 0.24 | 0.11 | ||||
| W1-2-1 | Pyrobitumen-clay composites | 0.42 | 0.16 | 0.25 | 0.06 | Highly developed pores with small pore diameter and micro-fractures associated with clay minerals, an areal porosity of 16.05%, | ||
| W1-2-2 | 0.27 | 0.16 | 0.23 | 0.15 | ||||
| W1-2-4 | 0.29 | 0.12 | 0.15 | 0.05 | ||||
| W1-2-5 | 0.25 | 0.10 | 0.14 | 0.04 | ||||
| Y1-67 | Homogeneous pyrobitumen | 0.29 | 0.23 | 0.38 | 0.19 | Poorly developed pores with non-uniform pore diameter and an areal porosity of 4.2%, | ||
| Y1-69 | 0.32 | 0.32 | 0.39 | 0.18 | ||||
| Y1-88 | 0.35 | 0.18 | 0.31 | 0.11 | ||||
| Y1 | 4 145.55 | Y1-68 | Pyrobitumen-clay composites | 0.25 | 0.15 | 0.13 | 0.08 | Highly developed pores with large pore diameter and areal porosity of 16.3% and17.6%, Figs. 4b, 5b |
| Y1-57 | 0.24 | 0.15 | 0.13 | 0.08 | ||||
| Y1-77 | 0.27 | 0.12 | 0.20 | 0.06 | ||||
| N1-40 | Homogeneous pyrobitumen | 0.25 | 0.23 | 0.24 | 0.28 | Poorly developed pores with small pore diameter and an areal porosity of 4.8%, | ||
| N1-42 | 0.17 | 0.35 | 0.10 | 0.22 | ||||
| N1-43 | 0.22 | 0.19 | 0.16 | 0.11 | ||||
| N1 | 3 591.84 | N1-70 | Homogeneous pyrobitumen | 0.23 | 0.22 | 0.18 | 0.11 | More pores with large pore diameter and an areal porosity of 13.4%, |
| N1-71 | 0.20 | 0.20 | 0.21 | 0.10 | ||||
| N1-72 | 0.18 | 0.23 | 0.13 | 0.12 | ||||
| N1-73 | 0.33 | 0.28 | 0.29 | 0.12 | ||||
| N1-50 | Pyrobitumen-clay composites | 0.13 | 0.30 | 0.09 | 0.12 | Highly developed pores with large pore diameter and an areal porosity of 16.2%, | ||
| N1-52 | 0.19 | 0.25 | 0.10 | 0.13 | ||||
| N1-56 | 0.27 | 0.30 | 0.20 | 0.20 |