Introduction
1. Geological features of deep to ultra-deep carbonate reservoirs, and demand on simulation experimental equipment
| Formation | Depth/m | Tarim Basin | Sichuan Basin | Fluid features | ||
|---|---|---|---|---|---|---|
| Temperature/°C | Pressure/MPa | Temperature/°C | Pressure/MPa | |||
| Shallow- medium | <4 500 | <115 | <54 | <125 | <67.50 | Meteoric water, connate sea water, formation brine, organic acid |
| Deep | 4 500-6 000 | 115-135 | 54-72 | 125-165 | 67.50-90.00 | Formation brine, organic acid, inorganic acid, formation thermal fluid, mantle-derived hydrothermal fluid |
| Ultra-deep | 6 000-7 500 | 135-165 | 72-90 | 165-200 | 90.00-112.50 | Formation brine, inorganic acid, formation thermal fluid, or mantle- derived hydrothermal fluid |
| Super-deep | >7 500 | >165 | >90 | >200 | >112.50 | |
2. Development of simulation experimental device
2.1. Research and development status of reservoir visual simulation experimental device
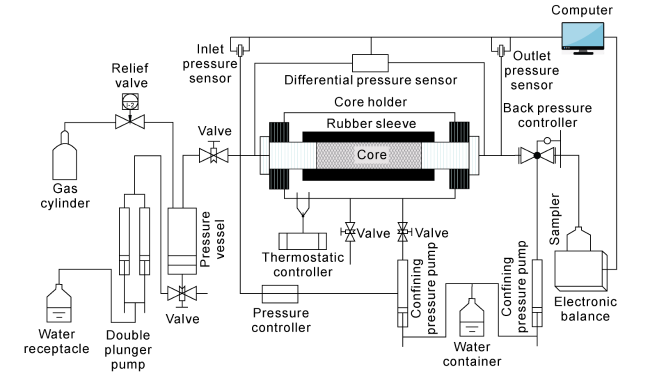
Fig. 1. HTHP core seepage simulation experimental device developed by CNPC Key Laboratory of Carbonate Rock Reservoir. |
2.2. Characteristics and functions of new reservoir visualization simulation experimental device
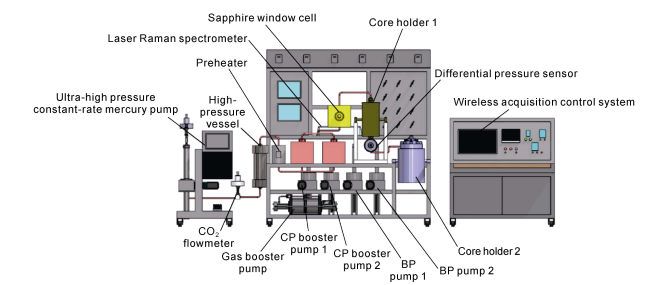
Fig. 2. Structure of the HTHP visualization simulation experimental device. |
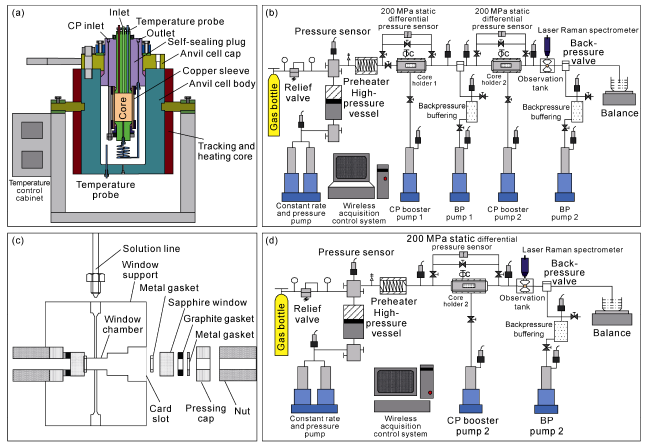
Fig. 3. Four functional modules of the HTHP visualization simulation experiment device. (a) Ultra-high temperature-pressure triaxial stress core holder and reactor module; (b) Multi-stage continuous flow module; (c) Internal parts of the ultra-high temperature -pressure sapphire window cell; (d) Real-time ultra-high temperature -pressure permeability detection module. |
2.2.1. Ultra-high temperature and high-pressure triaxial stress core holder module
2.2.2. Multi-stage continuous flow module with temperature and pressure adjustment function
2.2.3. Ultra-high temperature-pressure sapphire window cell and fluid real-time online detection module
2.2.4. Real-time online permeability detection module under ultra-high temperature -high-pressure
3. Simulation experiment on pore formation and preservation of deep to ultra-deep reservoir
3.1. Simulation experiment on the changes of pore-throat structure and fluid physical properties
Table 2. Experimental conditions for simulating the influence of lithology and initial pore-throat structure on dissolution effect in Tarim and Sichuan basins |
| No. | Samples lithology | Sample source | Porosity/ % | Permeability/ 10−3 μm2 | Experimental condition |
|---|---|---|---|---|---|
| 1 | Porous (vuggy) dolomite | Triassic Feixianguan Formation in the Sichuan Basin | 10.15 | 6.18 | 2 mL/L of acetic acid/CO2-saturated solution, open flow at 2 mL/min, dissolved inside. 8 TP points simulated in the Tarim Basin and 8 in the Sichuan Basin, representing 4 depths: shallow-medium, deep, ultra-deep and super deep. 60 min spent on each point |
| 2 | Fractured-porous (vuggy) dolomite | Cambrian Longwangmiao Formation in the Sichuan Basin | 10.80 | 32.40 | |
| 3 | Porous (vuggy) limestone | Ordovician Yijianfang Formation in the Tarim Basin | 7.08 | 0.70 | |
| 4 | Fractured limestone | Ordovician Yingshan Formation in the Tarim Basin | 2.79 | 3.30 |
Table 3. Results of dissolution simulation experiments on samples with different lithologies and initial pore-throat structures in Tarim and Sichuan basins |
| Sample No. | Physical properties at different temperature and pressure | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Initial physical properties | Shallow to medium | Deep | Ultra-deep | Super-deep | ||||||||||||||
| ϕ | K | 80 °C, 48 MPa | 110 °C, 66 MPa | 130 °C, 78 MPa | 150 °C, 90 MPa | 170 °C, 102 MPa | 190 °C, 114 MPa | 210 °C, 126 MPa | 230 °C, 138 MPa | |||||||||
| ϕ | K | ϕ | K | ϕ | K | ϕ | K | ϕ | K | ϕ | K | ϕ | K | ϕ | K | |||
| 1 | 10.15 | 6.18 | 10.20 | 6.2 | 10.32 | 6.25 | 10.45 | 6.38 | 11.05 | 8.65 | 12.15 | 13.15 | 13.27 | 15.23 | 13.67 | 18.4 | 14.95 | 21.2 |
| 2 | 10.80 | 32.40 | 11.12 | 46.7 | 11.44 | 64.30 | 11.69 | 79.60 | 11.89 | 95.70 | 12.05 | 110.50 | 12.15 | 131.50 | 12.25 | 149.7 | 12.30 | 182.4 |
| 3 | 7.08 | 0.70 | 7.30 | 27.5 | 7.48 | 112 | 7.63 | 207.00 | 7.77 | 323.00 | 7.87 | 587.30 | 7.99 | 876.50 | 8.04 | 1 467.0 | 8.08 | 1 650.7 |
| 4 | 2.79 | 3.30 | 2.90 | 69.0 | 2.98 | 189 | 3.03 | 268.00 | 3.07 | 367.00 | 3.13 | 604.00 | 3.19 | 973.00 | 3.23 | 1 143.0 | 3.29 | 1 278.3 |
Note that ϕ is porosity (%) and K is permeability (10-3 μm2). |
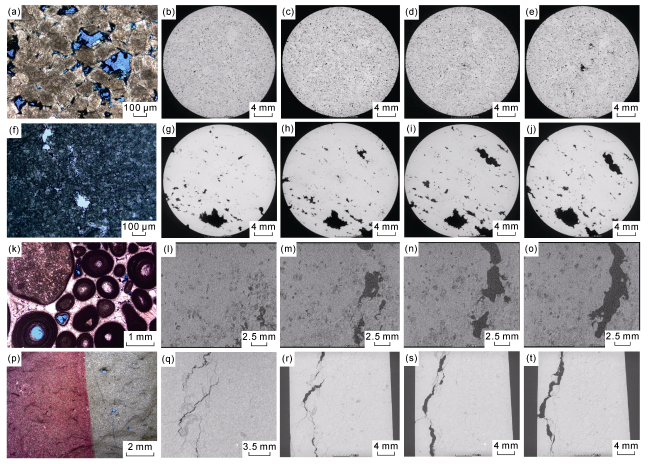
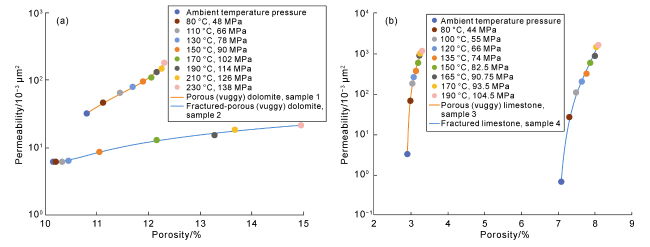
Fig. 4. Variation of pore-throat structure of different samples at different temperature and pressure points in burial and dissolution process. (a)-(e) Porous(vuggy) dolomite, Sample 1, with the conditions of cast thin section and plane-polarized light in (a), 80 °C and 48 MPa in (b), 130 °C and 78 MPa in (c), 170 °C and 102 MPa in (d), and 210 °C and 126 MPa in (e); (f)-(j) Fractured-porous(vuggy) dolomite, Sample 2, with the conditions of cast thin section and plane-polarized light in (f), 80 °C and 48 MPa in (g), 130 °C and 78 MPa in (h), 170 °C and 102 MPa in (i), and 210 °C and 126 MPa in (j); (k)-(o) Porous(vuggy) limestone, Sample 3, with the conditions of cast thin section and plane-polarized light in (k), 80 °C and 48 MPa in (l), 130 °C and 78 MPa in (m), 170 °C and 102 MPa in (n), and 210 °C and 126 MPa in (o); (p)-(t) Fractured limestone, Sample 4, with the conditions of cast thin section and plane-polarized light in (p), 80 °C and 48 MPa in (q), 130 °C and 78 MPa in (r), 170 °C and 102 MPa in (s), and 210 °C and 126 MPa in (t). |
Fig. 5. Variation of porosity and permeability of plunger samples at different temperature and pressure points in Sichuan Basin and Tarim Basin (porosity data from offline detection). (a) Variation of porosity and permeability at different temperature and pressure points in porous (vuggy) dolomite and fractured (vuggy) dolomite in the Sichuan Basin; (b) Variation of porosity and permeability at different temperature and pressure points in porous (vuggy) limestone and fractured limestone in the Tarim Basin. |
3.2. Simulation experiment on pore formation and preservation
Table 4. Conditions and results of simulation experiments for dissolution rate controlling factors and dissolution laws of carbonate rocks |
| SN | Simulation experiment | Sample No. | Porosity/ % | Permeability/ 10-3 μm2 | Experimental conditions | Experimental results | |
|---|---|---|---|---|---|---|---|
| Shallow to medium (<70 MPa, <120 °C) | Deep to ultra-deep (˃70 MPa, ˃120 °C) | ||||||
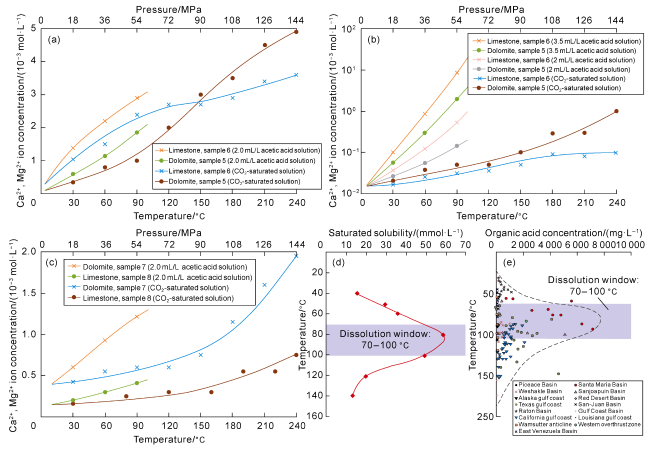
| 1 | Contrast experiment on dissolution rate for limestone and dolomite | 5 | 6.78 | 1.43 | 2 mL/L acetic acid/CO2-saturated solution; internal dissolution of rock; open flow at 1 mL/min | The dissolution rate of acetic acid to limestone was much higher than that to dolomite. With the increase of temperature and pressure, the dissolution ability of limestone and dolomite increased | With the increase of temperature and pressure, the dissolution rate of CO2- saturated solution to dolomite increased rapidly, and was evidently greater than that to limestone |
| 6 | 5.56 | 1.27 | |||||
| 2 | Effects of fluid type and concentration on dissolution rate of carbonate | 5 | 6.78 | 1.43 | 2 mL/L acetic acid solution, 3.5 mL/L acetic acid solution and CO2-saturated solution; internal dissolution of rock; open flow at 1 mL/min | The dissolution rate to carbonate increased with the increase of temperature and pressure. The dissolution rate of acetic acid was greater than that of CO2-saturated solution. The higher the concentration of acetic acid fluid, the greater the dissolution rate | The dissolution rate of CO2-saturated solution to dolomite increased rapidly and was evidently greater than that to limestone |
| 6 | 5.56 | 1.27 | |||||
| 3 | Effects of lithology and physical properties on dissolution rate of carbonate | 7 | 19.80 | 1.70 | 2 mL/L acetic acid solution and CO2- saturated solution; internal dissolution of rock; open flow at 1 mL/min | With the increase of temperature and pressure, the dissolution amount (ion concentration) of acetic acid solution and CO2-saturated solution to dolomite was greater than that to limestone | With the increase of temperature and pressure, the dissolution amount (ion concentration) of CO2-saturated solution to dolomite was greater than that to limestone |
| 8 | 4.40 | 3.60 | |||||
| 4 | Relationship between temperature and dissolution amount | 5 | 6.78 | 1.43 | 1 mL/L acetic acid solution, 2 mL/L acetic acid solution, 4 mL/L acetic acid, 7 mL/L acetic acid solution, 8 mL/L acetic acid solution; internal dissolution of rock; open flow at 1 mL/min | With the increase of temperature and pressure, the dissolution amount (ion concentration) of acetic acid solution to dolomite increased, and there existed a peak of dissolution | With the increase of temperature and pressure, the dissolution amount (ion concentration) of acetic acid solution to dolomite decreased |
Fig. 6. Simulation results of controlling factors on dissolution rate of various carbonate minerals. (a) Simulation experiment on the effect of carbonate mineral types on dissolution rate; (b) Simulation experiment on the effect of fluid type and concentration on carbonate rock dissolution rate; (c) Simulation experiment on the influence of lithology and physical properties on carbonate rock dissolution rate; (d) Simulation results of the relationship between temperature, organic acid concentration and dissolution amount of carbonate rock; (e) The relationship between organic acid concentration and formation temperature in global typical oil and gas basins/regions. |
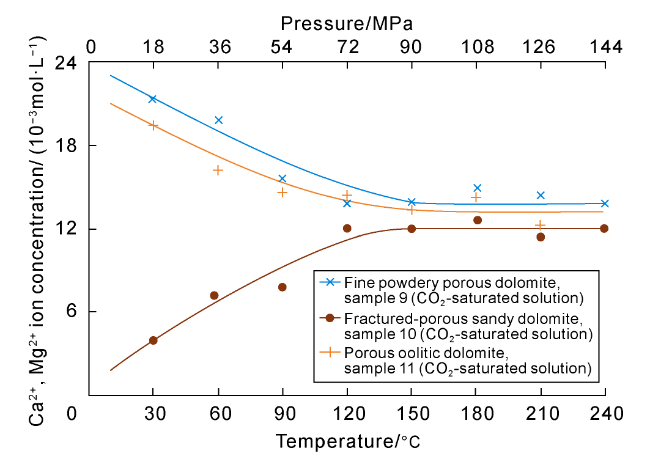
Fig. 7. Simulation experiments on pore preservation mechanisms of deep carbonate. Sample 9: Fine powdery porous dolomite, 5 994.00-5 994.15 m, Triassic Feixianguan Formation, Well LG001-1, Sichuan Basin. Sample 10: Fractured-porous sandy dolomite, 6 226.46-6 226.70 m, Triassic Feixianguan Formation, Well LG001-12, Sichuan Basin. Sample 11: Porous oolitic dolomite, 5 784.96-5 785.09 m, Triassic Feixianguan Formation, Well QB102, Sichuan Basin. |