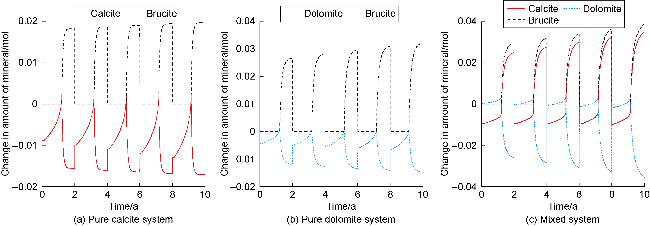
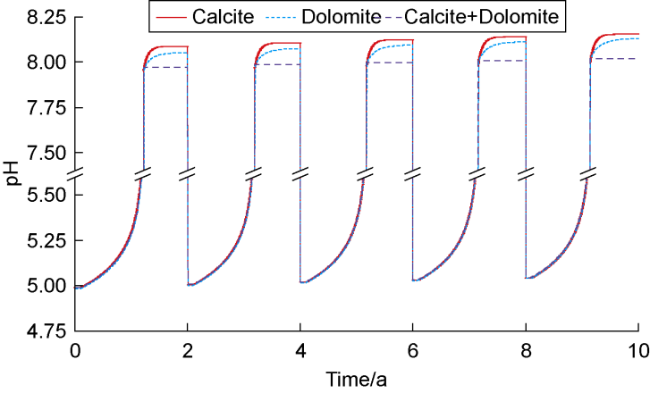
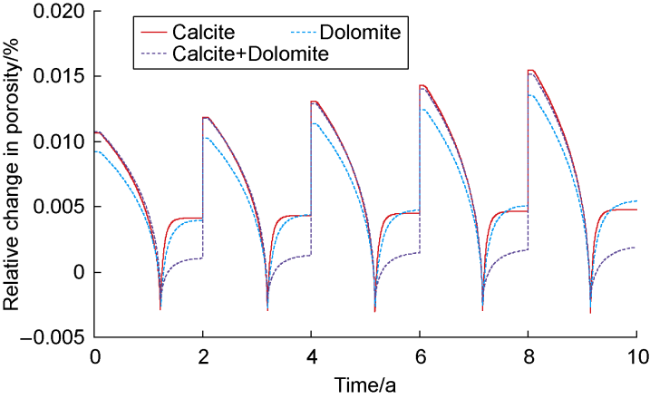
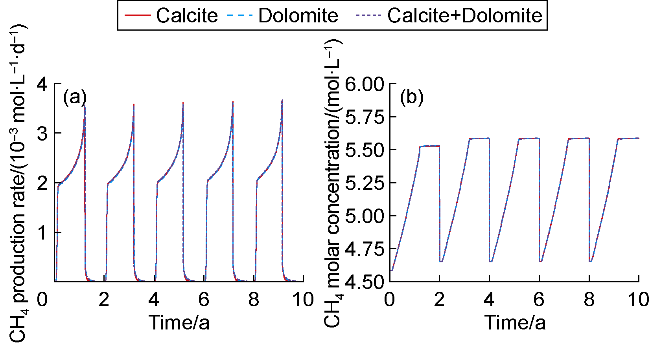
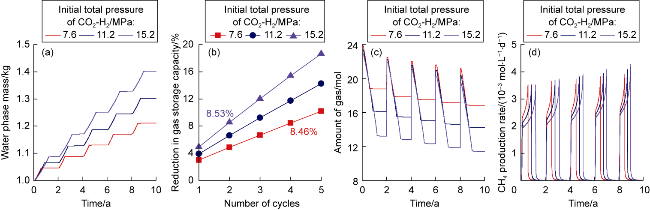
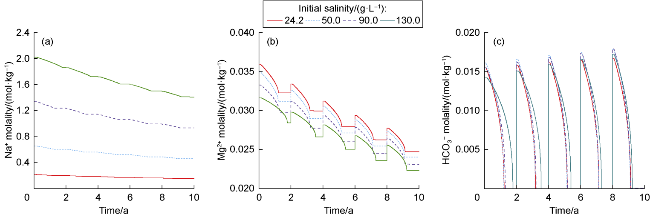
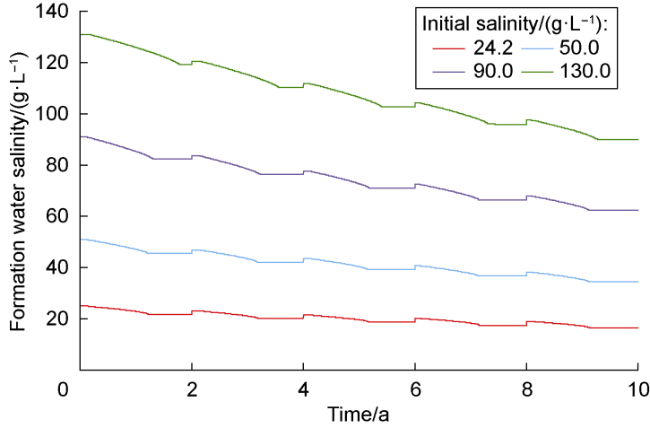
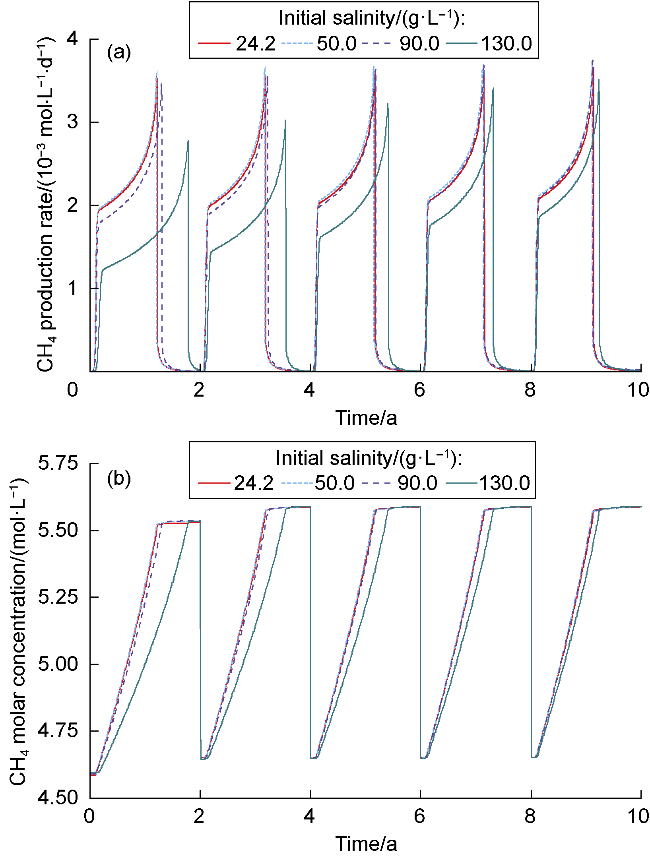
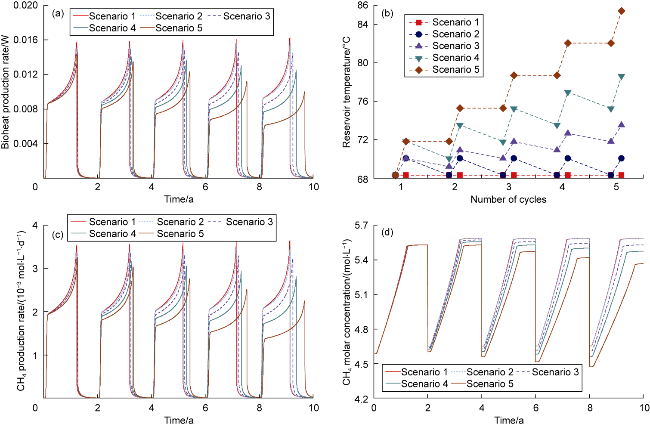
The changes in amounts of minerals during cyclic UBM under the different scenarios are shown in
Fig. 4, where positive values indicate mineral precipitation and negative values indicate mineral dissolution. In the pure calcite system, the high concentration of dissolved CO
2 in the early stage of the first cycle creates an acidic environment (
Fig. 5), triggering calcite dissolution. As methanogens continuously consume CO
2 and H
2 and produce water, the pH rises, reducing calcite dissolution and leading to slight precipitation. Once dissolved inorganic carbon (e.g., HCO
3− and CO
32−) in the formation water is depleted, calcite dissolves again to maintain carbonate equilibrium. During this process, the pH sharply increases, resulting in the precipitation of brucite (Mg(OH)
2), which stabilizes after the methanation reaction ends. In subsequent cycles, the dissolution and precipitation behaviors of calcite and brucite show periodic fluctuations. However, due to the cumulative increase in the water volume from methanogen metabolism, more calcite must dissolve each cycle to re-establish equilibrium. Additionally, the pH at the end of each cycle shows an increasing trend, causing the amount of brucite precipitation to incrementally increase (
Fig. 4a). In the pure dolomite system, the variation trend of amount of mineral is highly similar to that observed in the pure calcite system. Since dolomite dissolution supplies the Mg
2+ required for brucite precipitation, the amount of brucite precipitated at the end of the cycles in the pure dolomite system increases slightly compared to the pure calcite system (
Fig. 4b). In the calcite-dolomite mixed system, the trend of brucite precipitation is generally consistent with the previous two systems, whereas the behaviors of calcite and dolomite differ
[29]. During the pH gradual rising phase, ionization of H
2CO
3 leads to precipitation of both calcite and dolomite, causing calcite dissolution to gradually decrease and dolomite precipitation to increase. Subsequently, as the pH rapidly rises, brucite precipitation reduces the Mg
2+ concentration in formation water, inducing dolomite dissolution to replenish Mg
2+ levels while simultaneously raising Ca
2+ concentration, which in turn sustains continuous calcite precipitation. Through the combined actions of both minerals, brucite precipitation reaches approximately 0.039 mol after the fifth cycle (
Fig. 4c).