Introduction
1. Experiment introduction
1.1. Experimental samples
1.2. Experimental designs
Table 1. Experimental design for acid immersion in water- rich pores of volcanic rocks with different lithologies |
| SN | Lithology | Sample status | Type of acid solution | Porosity/ % | Permeability/ 10-3 μm2 |
|---|---|---|---|---|---|
| A1 | Breccia | Saturated with water | 10%HCl+1% HF | 7.62 | 0.318 5 |
| A2 | Dacite | Saturated with water | 10%HCl+1% HF | 6.42 | 0.129 5 |
| A3 | Andesite | Saturated with water | 10%HCl+1% HF | 7.23 | 0.389 0 |
| A4 | Tuff | Saturated with water | 10%HCl+1% HF | 7.35 | 0.293 5 |
Note: The experimental temperature is 25 °C and the pressure is 0.1 MPa. |
Table 2. Experimental design for acid immersion in oil- rich pores of volcanic rocks with different lithologies |
| SN | Lithology | Sample status | Type of acid solution | Porosity/ % | Permeability / 10-3 μm2 |
|---|---|---|---|---|---|
| B1 | Breccia | Saturated with oil | 10%HCl+1% HF | 7.83 | 0.331 2 |
| B2 | Dacite | Saturated with oil | 10%HCl+1% HF | 6.53 | 0.140 2 |
| B3 | Andesite | Saturated with oil | 10%HCl+1% HF | 7.26 | 0.386 2 |
| B4 | Tuff | Saturated with oil | 10%HCl+1% HF | 7.36 | 0.287 3 |
Note: The experimental temperature is 25 °C and the pressure is 0.1 MPa. |
Table 3. Experimental design of acid immersion in oil-rich pores of andesite under different temperature and pressure conditions |
| SN | Lithology | Sample status | Type of acid solution | Temperature/°C | Pressure/ MPa |
|---|---|---|---|---|---|
| C1 | Andesite | Saturated with oil | 10%HCl+ 1% HF | 70 | 30 |
| C2 | Andesite | Saturated with oil | 10%HCl+ 1% HF | 80 | 30 |
| C3 | Andesite | Saturated with oil | 10%HCl+ 1% HF | 90 | 30 |
1.3. Experimental methods
1.3.1. Acidification experiment
1.3.2. Characterization of mineral composition and pore structure
2. Results and analysis
2.1. Evolutionary characteristics of mineralogy and pore structure
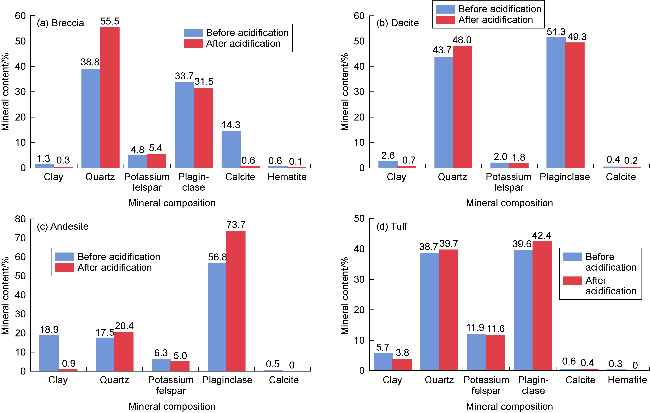
2.1.1. Changes in the mineral composition
Fig. 1. Changes in mineral composition of volcanic rock samples of different lithologies before and after acidification. |
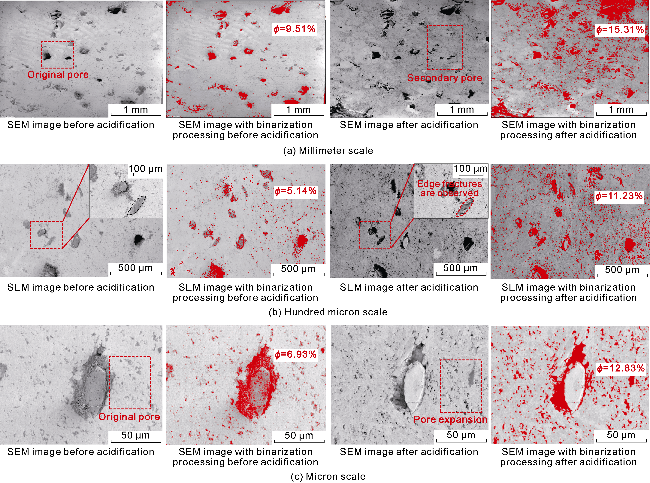
2.1.2. Changes in the pore structure
Fig. 2. Surface pore changes of tuff samples at multiple scales before and after acidification. |
2.2. Migration characteristics of acidic ions in water-rich pores
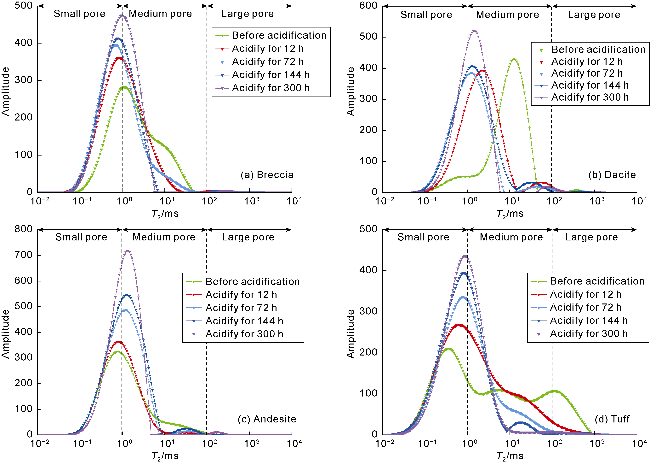
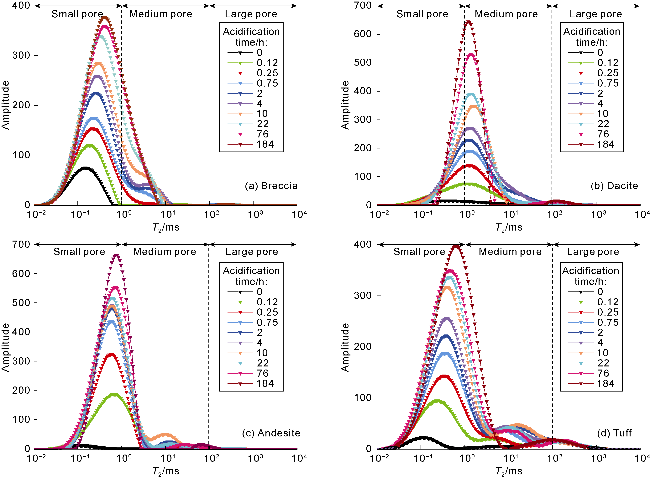
2.2.1. Analysis of NMR T2 spectra
Fig. 3. T2 spectra of water-rich pores in volcanic rocks of different lithologies before and after acidification. |
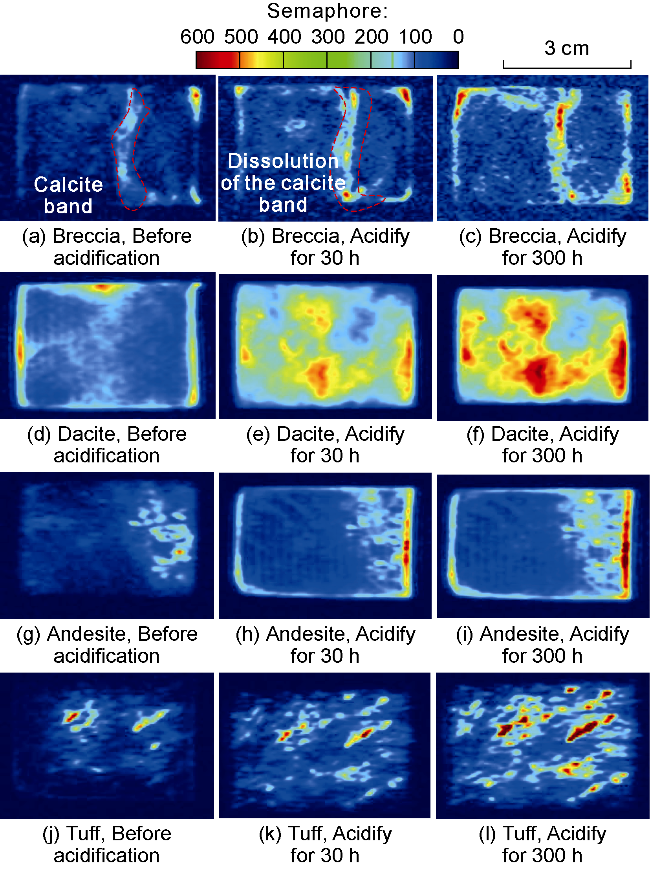
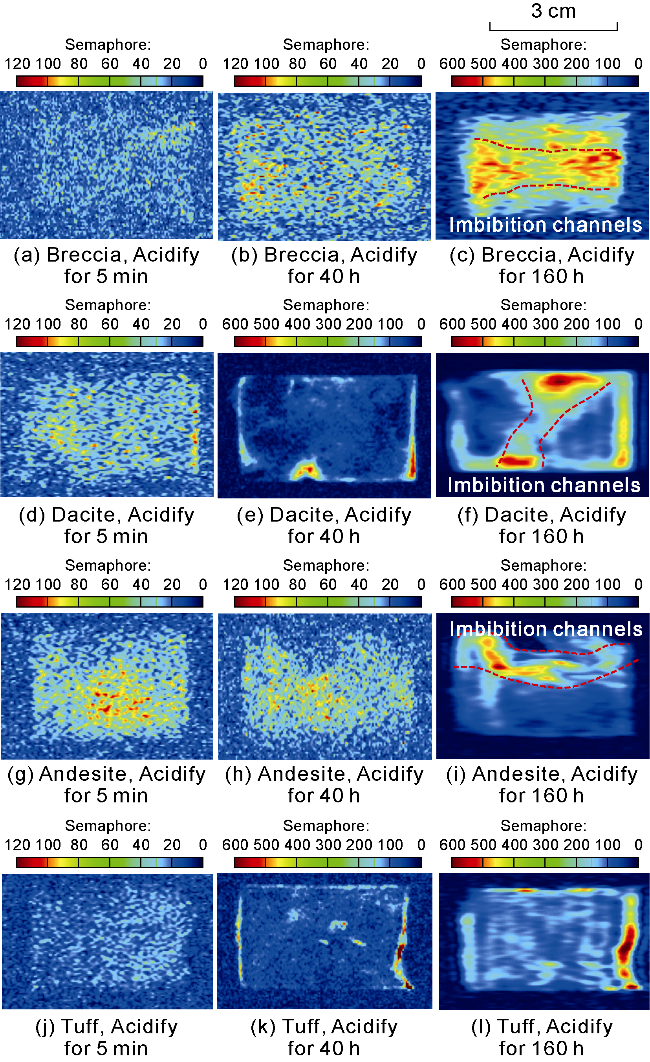
2.2.2. Analysis of MRI
Fig. 4. The MRI of water-rich pores in volcanic rocks of different lithologies before and after acidification. |
2.3. Characteristics of acid imbibition in oil-rich pores
2.3.1. Analysis of NMR T2 spectra
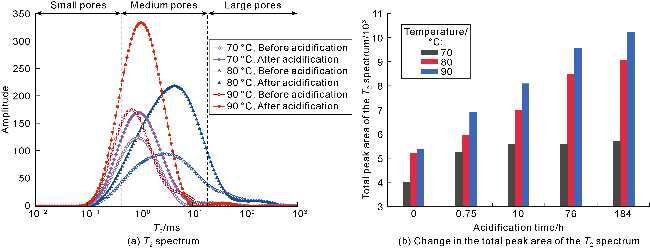
Fig. 5. T2 spectra of oil-rich pores in volcanic rocks of different lithologies before and after acidification. |
Fig. 6. Changes in the total peak area of T2 spectra of oil-rich pores in andesite before and after acidification under different temperature and pressure conditions. |
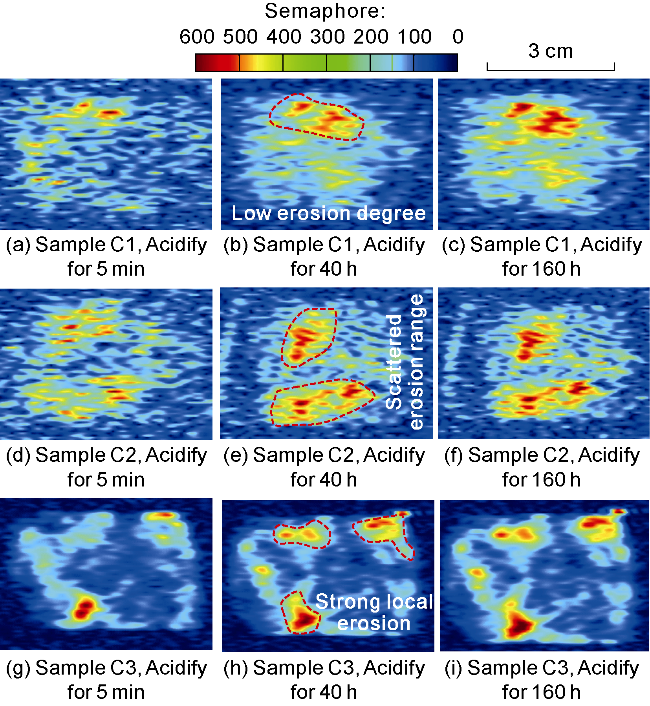
2.3.2. Analysis of MRI
Fig. 7. The MRI of oil-rich pores in volcanic rocks of different lithologies before and after acidification at normal temperature and pressure. |
Fig. 8. The MRI of oil-rich pores in andesite before and after acidification under high temperature and pressure. |
2.4. Correlation between H+ diffusion and acid imbibition
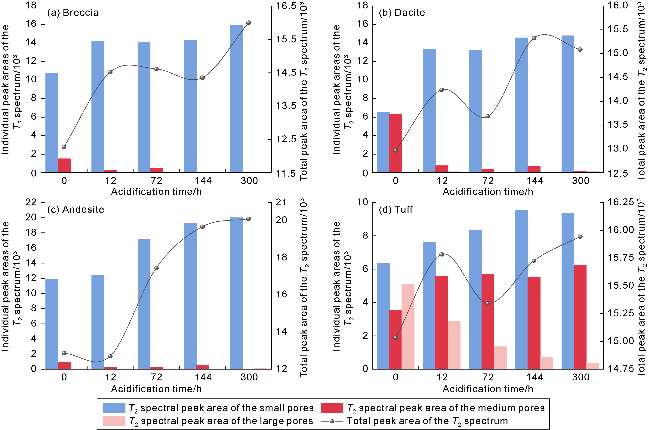
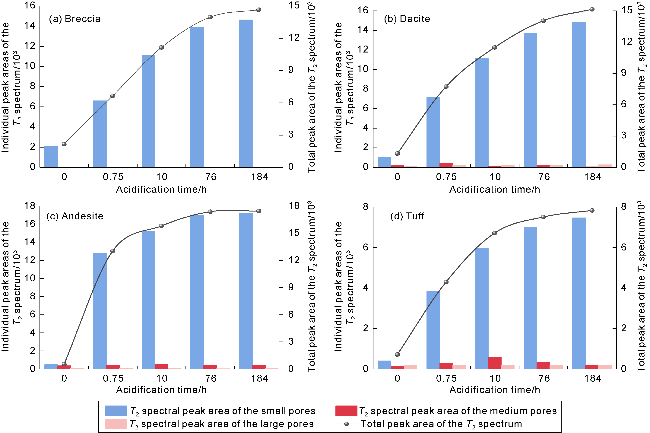
2.4.1. Erosion law of H+ diffusion and acid imbibition
Fig. 9. Total peak area and individual peak variations in the T2 spectrum during acidification of water-rich pores in volcanic rocks of different lithologies. |
Fig. 10. Total peak area and individual peak variations in the T2 spectrum during acidification of oil-rich pores in volcanic rocks of different lithologies. |
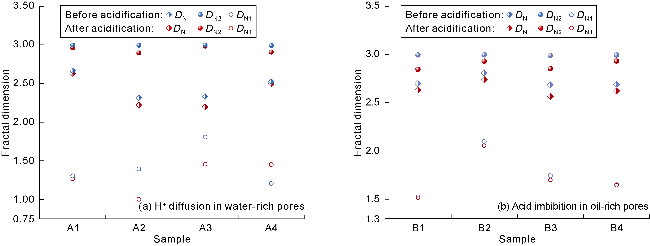
2.4.2. Mechanisms of H+ diffusion and acid imbibition
Fig. 11. Fractal dimension analysis of H+ diffusion in water-rich pores and acid imbibition in oil-rich pores of volcanic rocks. |
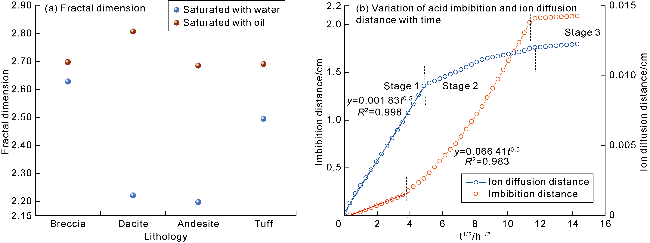
2.4.3. Correlation analysis of H+ diffusion and acid imbibition
Fig. 12. The fractal dimension of the water-rich and oil-rich pores of volcanic rock after acid erosion and the variation curve of the acid imbibition/ion diffusion distance with square root of time. |