Introduction
1. Current status of CCUS in major countries and regions
1.1. North American regions
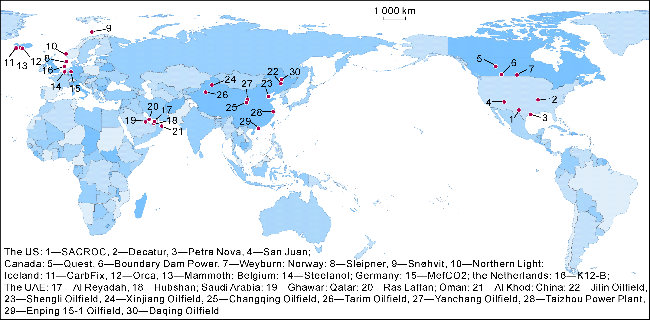
Fig. 1. Distribution of typical CCUS projects in the world. |
1.2. European region
1.3. Middle East region
1.4. China
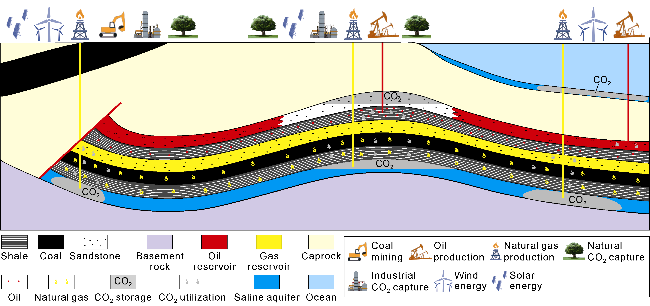
Fig. 2. CCUS model of oil and gas industry. |
2. CCUS technological progress
2.1. CO2 capture
| Capture method | Typical solvents/materials system | Operation system/°C | Energy required per 1 t CO2/GJ | Cost per 1 t CO2/ RMB | Capture efficiency/ % | Existing device scale | Technology maturity | Applicable scenarios |
|---|---|---|---|---|---|---|---|---|
| Physical adsorption | Zeolite, activated carbon, MOFs | 60-100 | 1.5-2.5 | 200-400 | 70.0-90.0 | Ten thousand tons class | Commercial application | Medium and high pressure gas, natural gas deacidification, hydrogen purification |
| Chemical adsorption | Amine solution (MEA, DEA, MDEA) | 85-120 | 2.0-4.0 | 300-500 | 80.0-95.0 | Million tons class | Commercial application | Large industrial flue gas capture such as thermal power, steel, cement and so on |
| Membrane separation | Polymer film, Inorganic film | <100 | 1.5-2.5 | 400-700 | 70.0-90.0 | Ten thousand tons class | Industrial demonstration | Small and medium-sized continuous separation such as natural gas deacidification, chemical tail gas treatment |
| Cryogenic separation | -140 to -70 | 4.0-7.0 | 500-800 | 95.0-99.9 | Ten thousand tons class | Industrial demonstration | Separation of high CO2 concentration associated gas, and purification of high pressure gas, natural gas, and hydrogen | |
| Direct air capture | Solid amine, strong alkali solution | 80-120 (Solid); 700-900 (Liquid) | 7.0-12.0 | 2 000- 4 000 | 60.0-90.0 | Ten thousand tons class | Industrial demonstration | Distributed, low concentration carbon capture |
| Chemical looping combustion capture | Metal oxides, oxygen carriers | 800-1 200 | 1.0-2.5 | 200-400 | 70.0-99.0 | Ten thousand tons class | Industrial demonstration | Integrated capture of combustion process |
2.2. CO2 utilization
2.2.1. CO2 geological utilization
2.2.2. CO2 chemical utilization
Table 2. Chemical conversion pathway of CO2 |
| Reaction type | Products | Chemical equation | Application direction |
|---|---|---|---|
| Hydrogenation | CO | CO2+H2→CO+H2O | Chemical raw materials, synthesis gas, metallurgical reducing agent, etc. |
| CH3OH | CO2+3H2→CH3OH+H2O | Liquid hydrogen storage, clean fuels, organic solvents, chemical intermediates, etc. | |
| HCOOH | CO2+H2→HCOOH | Chemical raw materials, hydrogen storage carriers, fuel cells, etc. | |
| CH4 | CO2+4H2→CH4+2H2O | Synthetic natural gas, clean fuel, chemical raw materials, etc. | |
| Dimethyl carbonate | CO2+3H2→CH3OH+H2O CO2+2CH3OH→CO(OCH3)2+H2O | Green solvents, battery electrolytes, gasoline additives, etc. | |
| Electrochemical reduction | HCOOH | CO2+2H++2e-→HCOOH | Chemical raw materials, hydrogen storage carriers, fuel cells, etc. |
| CH3OH | CO2+6H++6e-→CH3OH+H2O | Liquid hydrogen storage, electric drive CO2 conversion complementary to the thermal catalytic route | |
| Ethylene | 2CO2+12H++12e-→C2H4+4H2O | Plastic monomer, basic chemical raw materials, etc. | |
| Ethanol | 2CO2+12H++12e-→C2H5OH+3H2O | Renewable fuels, organic solvents, chemical intermediates, etc. | |
| Organic synthesis and polymerization reaction | Synthetic urea | 2NH3+CO2→(NH2)2CO+H2O | Fertilizer, urea-formaldehyde resin, feed additives, etc. |
| Polycarbonate | 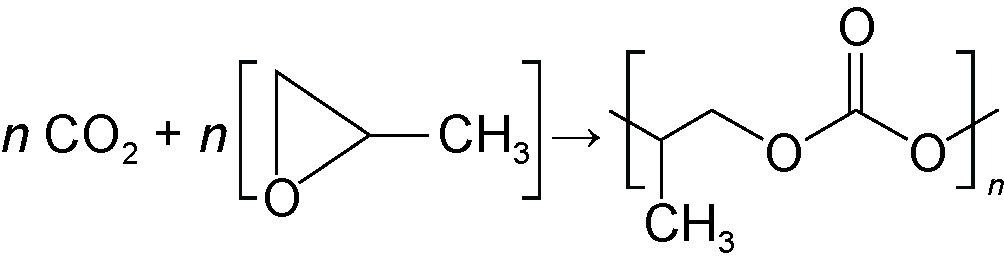 | Engineering plastics, biodegradable materials, etc. | |
| Cyclic carbonates | 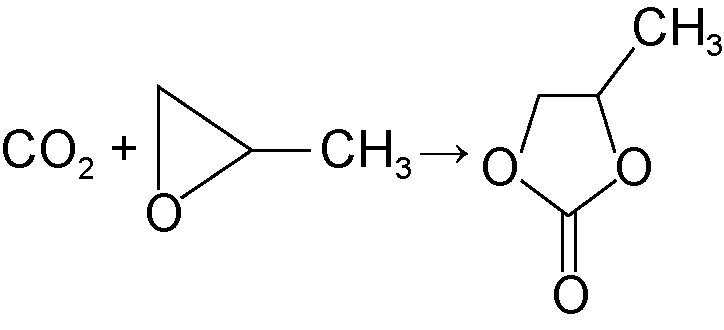 | Degradable plastic raw materials, battery electrolyte components, green solvents, etc. |
2.2.3. Other utilizations of CO2
2.3. CO2 storage
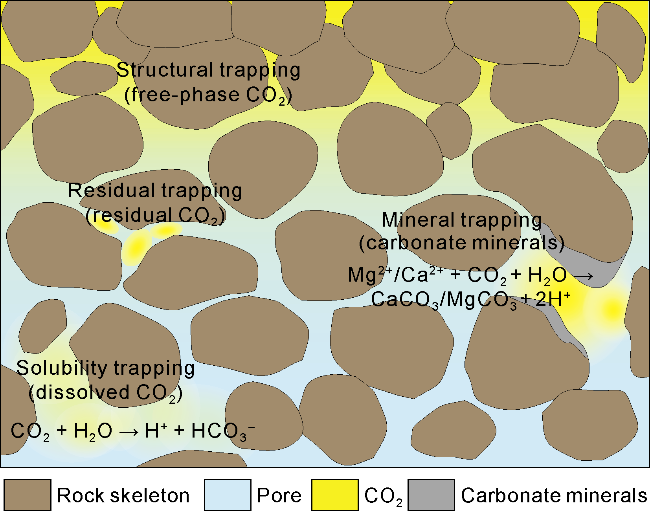
2.3.1. CO2 storage mechanism
Fig. 3. Microscopic schematic diagram of formation CO2 storage mechanism. |
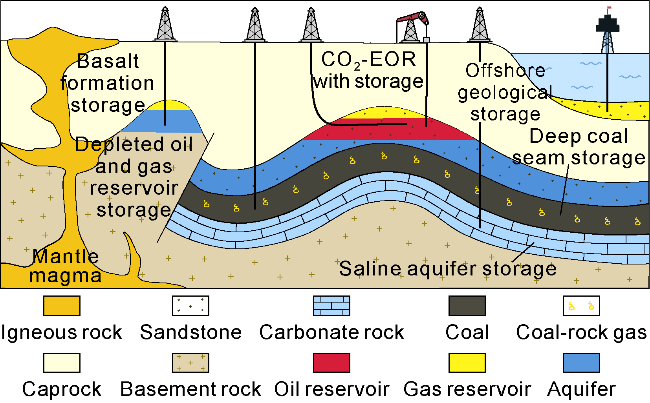
2.3.2. CO2 storaged geological body
Fig. 4. Geological storage of CO2. |