Introduction
Tight gas has become an important resource for global unconventional natural gas exploration and development [1]. Tight gas reservoirs show the characteristics of ultra-low porosity, ultra-low permeability, developed micro-fractures, strong capillary, rich clay minerals, and high possibility of formation damage. Laboratory test results show that the permeability of dry cores in tight gas reservoirs is more than 10 times than that of the original water-bearing cores, and the high original water saturation is one of the main factors that make it difficult for gas to flow [2]. Tight gas reservoirs have small pore throats and can be easily blocked by water. In the primary pores of the reservoir, clay minerals such as the illite-montmorillonite mixed layer cut micropores into much smaller pores, which increases the gas flow resistance. In addition, clay minerals are hydrophilic. Minerals may swell with water and block the micropores. Formation water occupies the pore space, which can further reduce the effective diameter of gas flow channels, resulting in flow resistance to increase rapidly, and significant water-blocking effect. This greatly limits the economic and efficient development of tight gas. So there is an urgent need to develop technologies which can effectively connect pores and reduce flowing resistance. At present, the commonly used methods for removing water lock can be roughly divided into two categories: physical removal and chemical removal. Physical removal methods include increasing production pressure difference, extending shut-in time, hydraulic fracturing and anti-blockage nano-emulsion etc. [3,4,5,6,7]. Chemical removal methods include gas injection, surfactant injection, alcohol injection and acidization [8,9,10,11,12]. Among them, the physical removal methods are unable to displace the formation water into the pore-throats, and consume a lot of energy. Conventional chemical removal methods are less effective and may bring serious pollution. Hence, it is very necessary to develop new removal methods.
The authors propose a new method to remove the water-blocking effect, which is the reservoir drying method by using a drying agent. This new method can quickly reduce the formation water near the wellbore through chemical reaction to reduce flowing resistance while consumes formation water. The paper describes the development, evaluation, injection and application of the drying agent.
1. Development and performance evaluation of the drying agent
1.1. Development and evaluation of drying agent
A qualified drying agent system should be easy to be injected. The relative molecular weight of the drying agent should be small, the hardness and particle size should also be small, and the physicochemical property is safe. The product of the drying process should be gas, and with no water and few solid materials. On the drying test device, the drying agent was studied based on the carbon group elements in the P zone and the alkaline earth metal elements in the S zone.
The drying agent is a nano-ionic carbide after dispersion and surface modification of a carbon group metal compound, which is mainly composed of carbon from the carbon group and cluster elements of IA, ⅡA and ⅢA. The drying agent ACHM is a mixture of two types of ionic carbon compounds, namely, C34- containing ionic carbide AC and C22- containing ionic carbide CHN [13,14]. The photo of actual product is shown in Fig. 1. AC reacts slowly when encountering cold water, but quickly and violently when encountering hot water, forming hydroxides and methane. Hydroxides are amphoteric hydroxides, which can react with acids or strong bases. CHN reacts quickly with water, releasing a lot of heat, generating soluble alkali and acetylene. The combination of AC and CHN plays a synergistic role in drying and basic salt resistance[14].
Fig. 1.
Fig. 1.
Photo of the drying agent.
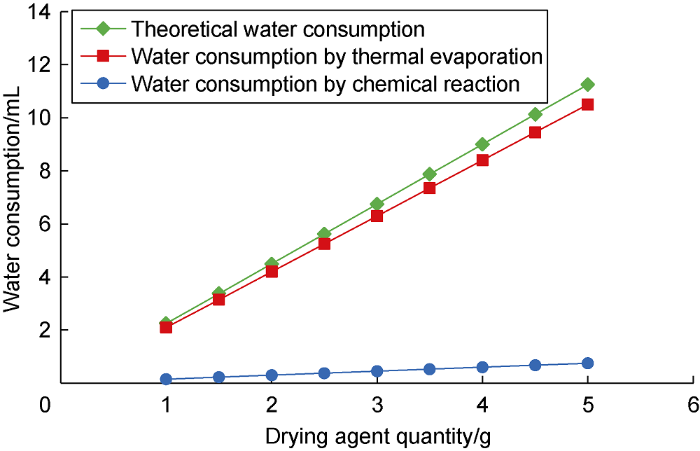
The reaction of 1 g of drying agent with formation water can release about 4.2 kJ of heat, and the thermal effect is significant. The reaction between AC and formation water is exothermic, with released heat accounting for about 95.9% of the total heat, while the synergist CHN reaction emits less heat. Fig. 2 shows the water consumption through thermal evaporation and total theoretical water consumption of drying agents with different quantity. The heat released by drying reaction can evaporate a large amount of formation water, and the water consumption accounts for about 70% of total theoretical water consumption, while the water consumption by chemical reaction of drying agent is relatively low [15,16].
Fig. 2.
Fig. 2.
Water consumption of thermal evaporation and total theoretical water consumption of different quantity drying agents.
In order to evaluate the performance of the drying agent, the evaporation-induced water consumption and total theoretical water consumption were measured at high temperature and high pressure. The results show that the drying agent has good temperature and pressure resistance characteristics. The effect of temperature and pressure on heat and water consumption is limited. The released heat varies with pressure and temperature by 0.002 8 kJ/MPa and 0.003 5 kJ/K, respectively [16].
1.2. Development and optimization of anti-salt agent
The primary drying component is mainly an ionic compound composed of P-zone carbon group elements, S-zone alkali metals, and alkaline earth metal elements. On the other hand, there are widespread calcium, magnesium, iron, lead, copper and manganese ions in formation water. So it is easy to salt out due to the products of drying process and the reduction of formation water [17,18,19]. LC, a compound composed of P-zone carbon group elements and S-zone alkali metal elements, has been studied and selected as a salt inhibitor. LC reacts with the reacting products of AC and CHN+water, and produces water-soluble NAO2 and LAO2, which can easily ionized into N+, L+ and AO2- in the remaining formation water, consequently reducing solid salt after drying reaction.
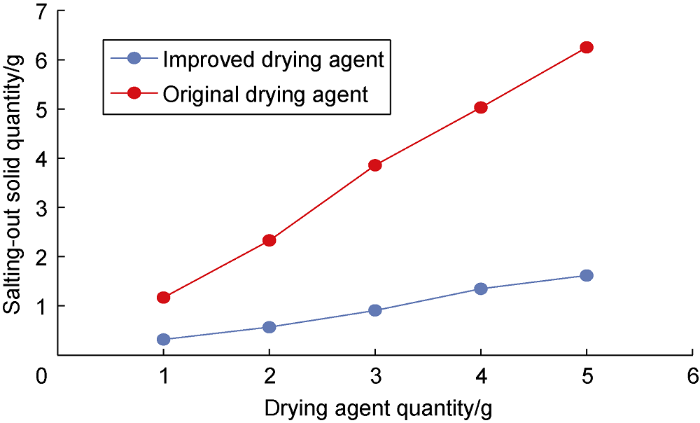
The anti-salt agent not only strengthens the thermal effect of the developed drying agent, but also significantly enhances its salt resistance, and further improves the ability to dry the connate water in reservoir [20]. The thermal effect increased from 4.2 kJ/g to 4.74 kJ/g, and the drying efficiency under the same thermal effect increased from 76.8% to 93.0%. After adding the anti-salt agent into the original drying agent, the salting-out solid produced by the unit mass of drying agent was reduced by about 70% (Fig. 3), and the effect became more significant as the drying agent increased.
Fig. 3.
Fig. 3.
Salt-resistance performance comparison.
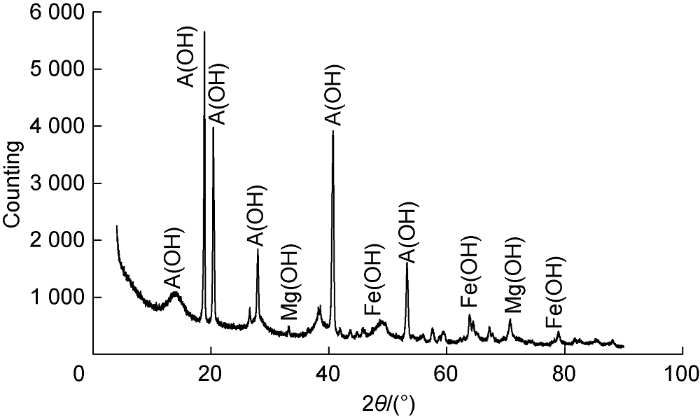
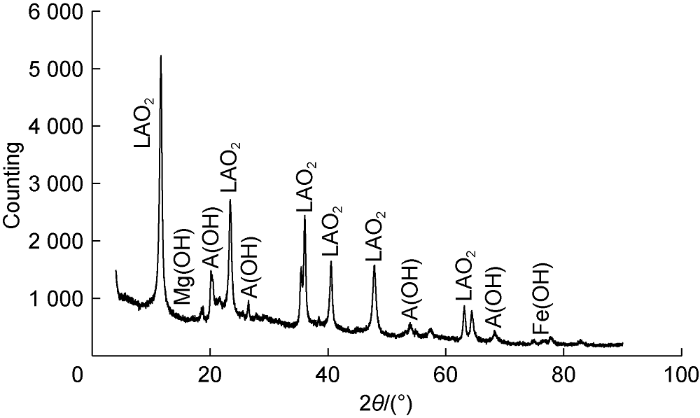
We compared the remaining salted-out products of the reactions between the original drying agent and the improved drying agent (adding the anti-salt agent LC) with formation water by X-ray diffraction phase analysis (Figs. 4 and 5). According to the phase composition at the absorption peak, the A(OH) solid component significantly reduced after adding the anti-salt agent LC, so did the measured amount of the salted-out products. The anti-salt agent LC obviously improved the salt-resistance of the drying agent.
Fig. 4.
Fig. 4.
X-ray diffraction result of salted-out products from original drying agent. θ—diffraction angle.
Fig. 5.
Fig. 5.
X-ray diffraction result of salted-out products from improved drying agent. θ—diffraction angle.
According to the X-ray diffraction peak shown in Fig. 5, after adding LC to improve salt resistance, the salting-out products still have solid composition from metal cations such as iron and magnesium. In this case, if adding a small amount of diethyltriamine-pentaacetic acid (DTPA) and ethylenediaminetetraacetic acid (EDTA), they can quickly react with calcium, magnesium, iron, lead, copper and manganese ions to form water-soluble complexes, which can further reduce salting-out products. These two agents are effective anti-salt adjuvants, and can further improve the salt-resistance of the drying agent [20].
1.3. Modification of drying agent
In the water-bearing tight gas reservoir at high temperature and high pressure, it is easy for the drying agent to lose reactivity, which makes it failiure in removinge water blocking. To meet injection requirements and delay drying reaction for tight gas reservoir, it is necessary to modify the drying agent. The solution polymerization method was used in this study. We used absolute ethanol as the reaction medium, under the action of the initiator of azobisisobutyronitrile (AIBN), and polyvinylpyrrolidone (PVP) for modifying the surface of ultra-fine AC powder. We then used the SEM (scanning electron microscopy), FTIR (Fourier transform infrared spectroscopy) and other methods to characterize the structure of the ultra-fine AC powder sample before and after treatment[21,22].
Due to the small particle size and high surface activation energy of ultra-fine AC powder, it can easily agglomerate during preparation and operation. The thermostatic magnetic stirring and dispersion treatment is employed to modify the ultra-fine powder. With the effect of the initiator AIBN, the ultra-fine AC powder after the treatment has a much more regular shape, and there is no smoothness and specular reflection phenomenon on the surface (Fig. 6).
Fig. 6.
Fig. 6.
SEM images of ultra-fine AC powder before and after treatment.
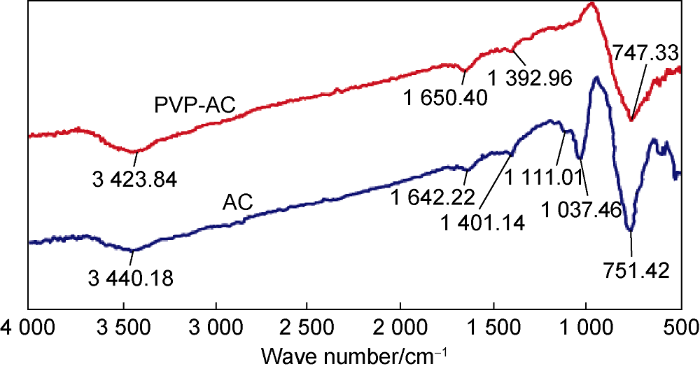
Fig. 7 shows that the ultra-fine AC powder modified through solution polymerization reaction has both the characteristic absorption peak of AC and the characteristic absorption peak of PVP. After the modification, the characteristic absorption peaks at 1037 cm-1 and 1111 cm-1 disappeared, indicating that the modification is effective. Comparing the characteristic absorption peaks of PVP, the C—N stretching vibration band at 1291 cm-1 and the band produced by C==O vibration transition at 1641 cm-1 have changed, which occurred at 1392 cm-1 and 1650 cm-1 respectively. The O-H stretching vibration absorption peak of 3370 cm-1 occurred at 3423 cm-1 after the modification.
Fig. 7.
Fig. 7.
FTIR spectra of ultra-fine AC powder before and after modification.
In Fig. 7, the characteristic absorption peaks of the polar groups on the surface of the ultra-fine AC powder moved toward the high wave number area after PVP modification, indicating new polar chemical bonds appear on the surface.
Delay test shows that the drying reaction time is delayed by 6-8 min after modification [23].
1.4. Methods for carrying drying agent into micro-nano pores
The pore size of tight gas reservoir is mainly in nanoscale ranging from 40 nm to 700 nm. The three compounds in the developed drying agents have relatively easy-to-prepare particle sizes, with a particle size of 1-10 μm, and there is currently no industrial nanoscale powder production method. Available drying agent powder can be used for drying natural micro-fractures and fracturing fractures. In order to dry the formation water in micro- and nano-pores in deeper layers, it is necessary to study how to carry drying agents into them. The agent dissolving and carrying method by supercritical CO2 was considered in the study. On the basis of an improved PVT (pressure-volume-temperature) device, a method for testing the solubility of the drying agent in supercritical CO2 was established [16, 20, 23-24], and preliminary research was conducted on basic thermodynamics [25,26,27]. The dissolution characteristics of the drying composition and synergist of the drying agent were tested by the phenomenological method with high-definition video and TV amplification. The dissolved substance and the original drying agent were compared and analyzed through energy dispersive spectroscopy (EDS) and X-ray diffraction (XRD) [22]. Since CHN is an alkali metal organic compound in the S zone, it must be dissolved in supercritical CO2, so only AC and LC need to be tested. After the interacting between AC and LC and supercritical CO2, the content and atomic percentage of the corresponding elements of the precipitates are basically the same as the original AC and LC, and the characteristic peak intensity is basically the same, showing the same structural characteristics [16, 20]. The elemental composition and structure of the precipitates are still the same as the original AC and LC, that is, the precipitated substance is the same as the original drying agent, and its chemical structure has not changed after dissolution. This indicates that the drying agent is soluble in supercritical CO2, so the drying agent can be dissolved and injected into reservoir [23].
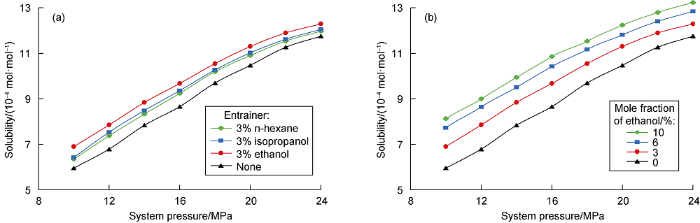
In order to increase the solubility of the drying agent, ethanol, isopropanol, and n-hexane were used as entrainers to test solubilization effect, and the best entrainer and its concentration were selected [16, 20] (Fig. 8). All the three kinds of entrainers can increase the solubility of the drying agent in supercritical CO2, but the increment is small. Among them, the solubilization effect of ethanol is the best, which is less affected by system pressure. The average lifting factor is 1.104. When supercritical CO2 is used to dissolve and carry the drying agent, ethanol should be used as an entrainer, and the mole fraction of the entrainer should be 10%.
Fig. 8.
Fig. 8.
Variation of solubility of drying agent in supercritical CO2 with system pressure at 40 °C under conditions of different types of entrainers (a) and different mole fractions of ethanol (b).
2. Test and evaluation of drying performance
We investigated the drying performance within the micropores and the types of dried water using etched-glass model based on tight reservoir. Then, we conducted rock test to evaluate the drying performance in combination of NMR technique [28].
2.1. Testing and evaluation of microscopic drying performance
The microscopic visual model of tight sandstone was constructed by laser engraving technique. In this model, the pore structure was extracted from casting thin section of tight sandstone, and laser engraving technique was applied to etch the pore structure on the glass model, with equal proportion of the extracted one. The drying performance affected by microscopic structure can be observed, and the drying agent can be qualitatively and semi-quantitatively evaluated [29]. The model and the corresponding visualizing system are shown in Fig. 9.
Fig. 9.
Fig. 9.
Visual model and imaging system.
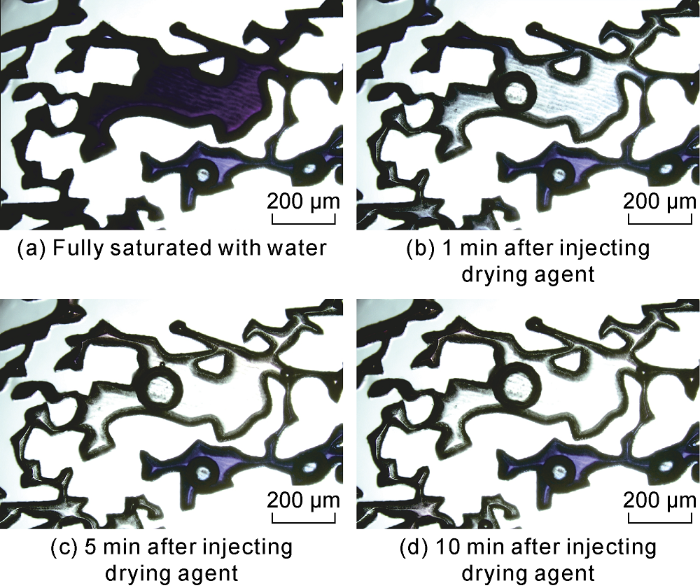
Under three temperature conditions (40 °C, 50 °C and 60 °C), the drying process of ethanol-carried drying agent particles in the etched model was revealed. Good drying performances were observed at these temperatures. Fig. 10 shows the drying process at 60 °C [29]. At these temperatures, different sizes of gas bubbles created from the chemical reaction between the drying agent and water were observed in the model, and sometimes the bubbles even formed continuous phases. The bubbles were able to displace thin film water and water in dead ends, then enhancing drying performance [29]. In addition, the drying reaction is very sensitive to temperature. Higher temperature is much more helpful, especially in the model with high water saturation.
Fig. 10.
Fig. 10.
Drying process at 60 °C (color area represents water phase).
By comparing and analyzing the water distribution within the etched model before and after drying (Figs. 11 and 12), the drying efficiency was calculated through image and statistical analysis. In this case, the drying efficiency is very high (51.43%).
Fig. 11.
Fig. 11.
Residual water distribution within the microscopic etched model before drying.
Fig. 12.
Fig. 12.
Residual water distribution within the microscopic etched model after drying.
The microscopic etched model can be used to compare the effect of different pore structures on the drying efficiency. As shown in Table 1, the drying efficiency of pore structures such as flaked, curved throats is better than that of spot-shaped ones. The drying efficiency of the residual water phase in flaked and curved throats is up to 95%. Considering the distribution characteristics of residual water, the main reason is that residual water mainly exists as thin films in flaked throats, so the drying agent can fully contact with them and trigger drying reaction.
Table 1 Drying efficiency on different pore structure models
| Pore structure model | Surface porosity/% | Irreducible water saturation/% | Water saturation after drying/% | Drying efficiency/% |
|---|---|---|---|---|
| Spot-shaped throats | 30.98 | 45.58 | 24.21 | 46.89 |
| Flaked/curved throats | 17.77 | 41.14 | 1.70 | 95.87 |
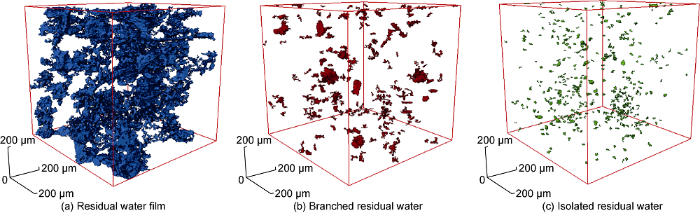
Using CT and digital core technology, the 3D distribution of residual water after drying was reconstructed. According to the irreducible water after gas flooding on the CT image, the residual water can be classified into three types (residual water film, isolated residual water and branched residual water) based on the sphericity coefficient and Euler number. The spatial distribution of the three types of residual water was reconstructed, respectively (Fig. 13). Therefore, the reservoir storage space containing residual water consists of five components: blind pore, residual water film, isolated residual water, branched residual water, and connected pores.
Fig. 13.
Fig. 13.
Residual water in a microscopic digital core.
Digital core in combination of lattice Boltzmann method (LBM) were used to simulate the gas permeability when drying different forms of residual water, and the effects of different forms of residual water on gas permeability were compared. The results show that residual water film has the greatest influence on gas permeability, followed by branched residual water, and isolated residual water has the least influence [30,31].
Furthermore, the drying influence of different forms of residual water on gas permeability was simulated on digital cores (Table 2). Residual water film has the greatest influence on drying efficiency, up to 98%.
Table 2 Drying effects of residual water on gas permeability
| Model with residual water | Pore structure | Improved gas permeability/% | |||
|---|---|---|---|---|---|
| Average pore radius/μm | Average throat radius/μm | Pore/throat ratio | Average coordination number | ||
| Dry core | 17.975 3 | 9.485 9 | 1.894 9 | 3.287 5 | |
| Branched residual water | 22.047 6 | 12.464 2 | 1.768 9 | 2.491 7 | 5.24 |
| Isolated residual water | 22.155 6 | 12.502 1 | 1.772 2 | 2.459 1 | 4.70 |
| Residual water film | 17.868 0 | 9.346 8 | 1.911 7 | 3.298 4 | 97.87 |
2.2. Evaluation of drying performance of porous media in different reservoirs
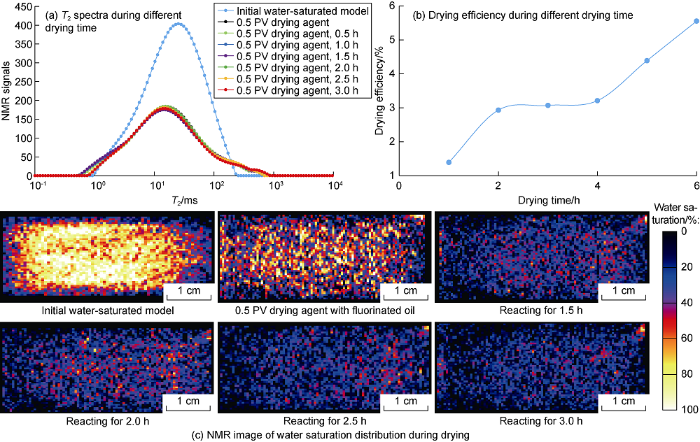
The supercritical CO2 system (supercritical CO2 as a carrier, and ethanol as an entrainer) contains ethanol and other substances. When evaluating the drying performance of the main component of drying agent by NMR technology, to avoid the interference from other substances in the supercritical CO2 system, fluorinated oil (without nuclear magnetic signal) was used as a carrier to prepare AC solution. The AC solution was injected into the core at 0.01 mL/min to quantitatively evaluate the drying performance of the main component of drying agent in porous media. By comparing core NMR images and T2 (transverse relaxation time) spectra in different drying durations (Fig. 14), it can be seen that the drying agent can effectively dry the water phase in the matrix pores [28], but the drying efficiency is not good (only 5%) due to low solubility of fluorine oil in water.
Fig. 14.
Fig. 14.
Drying performances and water distribution in tight cores dried by drying agent with fluorinated oil. PV - injected pore volume.
To capture the distribution and amount of water NMR signals, it is necessary to delimitate ethanol NMR signals during the dry process by supercritical CO2 system. Firstly, inject the drying agent system with ethanol suspension into a porous medium to trigger drying reaction, and detect the NMR signal of ethanol in the ethanol-water mixed fluid. Then inject the supercritical CO2 system to study the drying reaction process. Based on the ethanol NMR signal during ethanol suspension injection, the influence of ethanol NMR signal in the supercritical CO2 system could be removed, and then the distribution and amount of water NMR signals in the drying process of the system could be obtained.
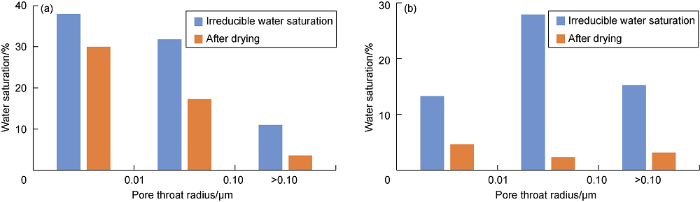
Comparing the experimental results of ethanol suspension injection and supercritical CO2 solution injection, it is found that the drying agent has a good drying performance on the residual water in both the large and small pore throats of tight core. In comparison, the performance of the drying agent solution with supercritical CO2 as a carrier is obviously better than that with ethanol (Fig. 15). Supercritical CO2 can effectively dissolve the main agent AC so that it can be transported into smaller pores and interact with the residual water in these pores. In this way, water in smaller pore throats can be effectively dried, and the throats become additional flow pathways for gas.
Fig. 15.
Fig. 15.
Comparison of water drying performance in different size of pore throats during ethanol suspension injection (a) and supercritical CO2 solution injection (b).
Tight cores with fractures and without fractures were used to conduct drying experiments and to evaluate the drying performance on porous media in different reservoirs. Before the experiments, irreducible water saturation of the cores was established using a gas drainage method. High-temperature and high-pressure flow experiment device equipped with online NMR was adopted, and the experiment steps are shown in Fig. 16. The experimental conditions include: 20 MPa confining pressure, 16 MPa displacement pressure, 10 MPa outlet back pressure, temperature of 90 °C, and reaction time of 120 mins.
Fig. 16.
Fig. 16.
Work flow of high temperature and high pressure drying experiments under jointing action of flow displacement and NMR technique.
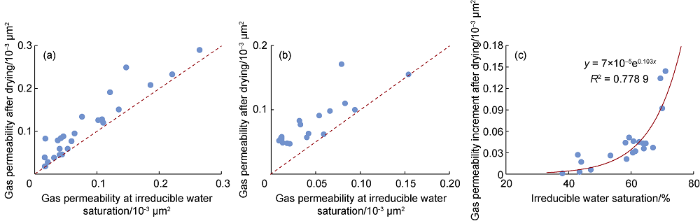
The experimental results show that, before injecting ethanol-carried drying agent, the average gas permeability of the tight core at irreducible water saturation is 0.078× 10-3 μm2, while the gas permeability increased to 0.107×10-3 μm2 after drying, with an average 9.63% reduction of water saturation in the core. When the supercritical CO2 dissolved drying agent was injected into the core, the average gas permeability of tight core increased from 0.049×10-3 μm2 (before drying) to 0.093×10-3 μm2 (after drying), with an average 16.21% reduction of water saturation in the core. It can be seen that both ethanol suspension and supercritical CO2 as carriers can effectively reduce the water saturation of tight reservoir cores and improve gas flowing. Their drying and flowing resistance reduction abilities are far better than removing water lock by heating or injecting supercritical CO2 (Table 3) [32,33]. In addition, there is an obviously positive correlation between the increment of gas phase permeability and bounded water saturation after drying when using supercritical CO2 as a carrier (Fig. 17). In other words, the higher the bounded water saturation is, the better the drying performance is.
Table 3 Comparison of physical properties of tight cores before and after drying
| Drying experi- mental groups | Average reduction in water saturation/% | Average increase of gas permeability/% |
|---|---|---|
| Heating control group | 3.07 | 13.60 |
| Supercritical CO2 control group | 5.19 | 25.70 |
| Ethanol suspension + drying agent | 9.63 | 54.38 |
| Supercritical CO2 + drying agent | 16.21 | 163.10 |
Note: the increase rate of gas permeability is calculated by subtracting the gas permeability at irreducible water saturation from the gas permeability after drying, and then the result divided by the gas permeability at irreducible water saturation. The final permeability increase rate is the average of all cores.
Fig. 17.
Fig. 17.
Comparison of gas permeability before and after drying by ethanol suspension injection (a) and supercritical CO2 solution injection (b); and the relationship between gas permeability increment and bounded water saturation after supercritical CO2 solution injection (c).
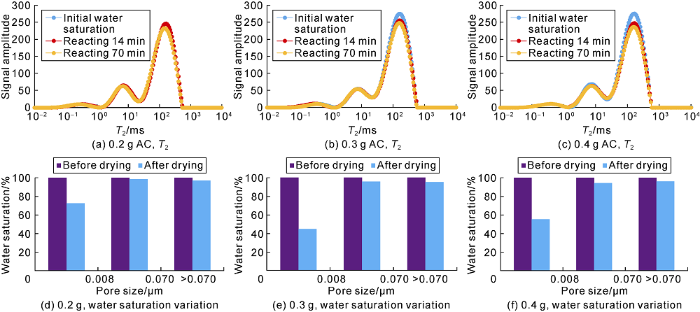
Under the laboratory temperature and pressure, the drying agent was placed on the fracture face to simulate the agent invading into the fracture during displacement, and the drying performances of the water in the fractures and in the near-fracture matrix pores were tested. The drying efficiency testing results of the three tight cores with fractures are shown in Table 4. Different amounts of drying agent AC were evenly placed on the fracture surface, and the changes of T2 spectra and water saturation during the drying process were measured. Fig. 18 shows the test results of No. 9 core, and the results of the other two cores are basically similar (not listed here). The T2 spectrum area gradually decreases and the water saturation becomes smaller with the process of drying reaction. The larger the quantity of the drying agent, the better the drying effect. In the drying process of tight cores with fractures, the drying agent is effective for all pores, but more effective in the large pores, while less effective in the small pores. This indicates that supercritical CO2 as a medium to carry drying agent is necessary for it can push the drying agent into small pores.
Table 4 The tested results of drying efficiency on tight cores with fractures
| Core No. | Gas porosity/ % | Gas per- meability/ 10-3 μm2 | Drying efficiency/% | ||
|---|---|---|---|---|---|
| 0.2 g AC | 0.3 g AC | 0.4 g AC | |||
| 313 | 26.74 | 6.850 | 3.88 | 4.80 | 4.88 |
| 780 | 28.76 | 39.630 | 4.51 | 6.43 | 9.23 |
| 9 | 6.43 | 0.364 | 8.13 | 11.64 | 12.90 |
Fig. 18.
Fig. 18.
Changes of T2 spectra and water saturation of fractured core (No. 9).
3. Effects of reservoir drying on gas flow
3.1. Microscopic simulation of reservoir drying
Different from conventional sandstone gas reservoirs, tight sandstone gas reservoirs experienced strong compaction and cementation during accumulation, resulting in narrow flow channels. Laboratory experimental results showed that the diameter of main flow channels is less than 100 nm, and gas flow exhibits obvious micro-scale effect.
Considering the effects of slippage effect and boundary Knudsen layer, a micro-scale flow simulation model for unconventional gas was established based on the lattice Boltzmann method (LBM). The micro-scale gas flow behavior and its influencing factors within the pore size of main flow channels in tight sandstone were studied, and the flow characteristics of weak continuous flow, slippage flow and weak transition flow were analyzed [34,35]. The results show that the size of flow channels and Knudsen number (Kn) are main controlling factors of gas flow, and the presence of throats makes gas pressure change nonlinearly, which greatly reduces gas flow capacity and mass flow [34]. The above conclusions reveal the importance of “opening fractures and expanding throats” for developing tight gas, and indicate that the water in throats should be removed during drying process. It is consistent with the visualized drying experiment using supercritical CO2.
The results of laser etched model and NMR online drying experiment show that the formation water can be reacted and consumed uniformly with the drying agent[29]. In LBM simulation, the drying effect was simplified as the consumption of formation water, and the improvement of tight gas flow capacity under different drying intensity and micro pores was studied. The results show that drying reservoir can effectively consume the water films on pore walls, the water trapped in connected throats, and the water in dead pores. This can significantly increase the gas flow area and improve the gas flow capacity. In addition, it is found the high-speed flow occurs in the narrow throats connecting with large pores. This also indicates that throats are an important part to dry. The existence of trapped water and water film will form an obvious water lock region, and the increase of drying intensity can further eliminate formation water, thus effectively improving the gas flow capacity.
3.2. Effect of drying effect on gas well productivity
The purpose of injecting drying agent is to consume the formation water in the channels near the wellbore while making pore channels originally occupied by formation water can be effective for gas flow to increase gas production. Previous experiments show that the drying effect is related to the amount and injection depth of drying agent. Based on abundant laboratory experimental data, an empirical relationship between gas permeability with injected amount and injection depth of drying agent was obtained. In addition, with narrow flow channels in tight sandstone, gas molecules may collide with pore wall to induce slippage effect; the effects of stress sensitivity and high-speed non-Darcy flow near the wellbore on productivity cannot be ignored. Based on these complex flow mechanisms, the productivity models of vertical and horizontal wells for developing tight gas reservoirs were established, and the drying effect was coupled with the productivity model based on empirical gas permeability equation. Finally, the drying effects (at different injected volumes and depths) were analyzed on the improvement of gas well productivity in various types of wells for tight gas reservoirs [16, 20].
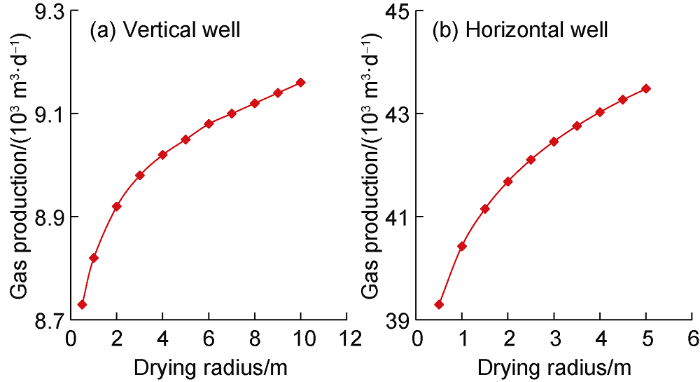
Fig. 19 shows the relationship between gas well productivity and effective drying radius (referred to as drying radius) when bottom hole flow pressure is 20 MPa. It is obvious that the gas well productivity increases with the increase of the drying radius. When the drying radius is less than 2 m, the productivity quickly increases with the increase of drying radius. In addition, by keeping a 2 m drying radius, the relationship between the amount of drying agent injected and the productivity was studied. It was found that increasing drying agent could effectively improve well productivity. In the vertical well, when the injected drying agent is less than 3 kg/m3, there is a quick increase of productivity. In the horizontal well, the critical amount is 1.5 kg/m3 [20].
Fig. 19.
Fig. 19.
Relationship between gas well production and drying radius at bottom hole flow pressure of 20 MPa.
3.3. Drying effect on the production performance of complex fractured wells
Compared with vertical wells, multistage fractured horizontal wells have better production performances. The fracturing stimulation is an effective technology for tight reservoir development. Hydraulic fracturing operation will induce asymmetrical and irregular hydraulic fractures with complex shape around the horizontal wellbore. Matrix pores, natural fractures and hydraulic fractures appear in the same reservoir, and they may induce fluid channeling. So it is of great significance to study the flow models for production performance prediction. The fluid flow characteristics of tight gas reservoirs and complex influencing factors (e.g. stress sensitivity) were considered, and an embedded discrete fracture model (EDFM) was used to characterize the complex fractures. Then a three-dimensional multiphase flow model of fractured horizontal wells for tight gas reservoirs was established, which was used as the basis of numerical simulation on drying effect [36].
Based on the principle of EDFM, a projection-method- based fracture generation technology and a numerical algorithm were established. A fracture network was obtained through the projection plane of fracture boundary and matrix grid system on each coordinate plane. A multiphase flow numerical simulator based on EDFM was developed to simulate the pressure dynamics and gas dynamics in the complex fracture network of the horizontal well in tight gas reservoir [37]. Simulation results show that the decrease of water saturation can effectively improve gas production and stable production capacity [37]. Due to severe water-lock effect around the wellbore, drying formation water will inevitably improve gas production performance.
4. Conclusions
For developing tight gas reservoirs, drying reservoir is a feasible method to improve the seepage capacity within the well-controlled area. Phase behavior test, microscopic analysis and core test have shown good drying performance of the proposed drying agent and the synergistic salt inhibitor developed on the chemical elements in S and P zones. The drying reaction is less affected by high temperature and pressure. The production of tight gas reservoir can be increased by injecting supercritical CO2 solution and delaying drying reaction.
Microscopic test and digital core simulation show that the drying effect can be over 90% for the water film in microscopic pore throats, especially the water in flaked throats. Both ethanol suspension and supercritical CO2 solution as carriers of drying agent have good drying efficiency on tight cores. The average decrease of water saturation using ethanol suspension as a carrier is 9.63%, and the core permeability under irreducible water conditions increases by 54.38%. The average decrease of water saturation using supercritical CO2 solution as a carrier is 16.21%, and the permeability under irreducible water condition increases by 163.1%.
Drying out water films and trapped water make pores connect with each other, and improve gas flowing through micropores. Both macroscopic productivity evaluation and numerical simulation results show that drying reservoir can greatly improve gas production and stable production capacity of the tight gas well.
The study on drying tight gas reservoir to enhance gas recovery mainly focuses on the formation water in pore throats and the water during lost circulation. The drying agent does not react with the reservoir itself. Different tight gas reservoirs have different pore structures and different salinity of formation water, so the drying efficiency and salt resistance have certain differences. However, the proposed method in this work could be modified to adapt different reservoir environment.
Reference
“Extreme utilization” development theory of unconventional natural gas
Mechanism and control factors of water blocking in tight sandstone gas reservoir
DOI:10.1360/N092017-00200 URL [Cited within: 1]
Spontaneous removal behavior of water phase trapping damage in tight sandstone gas reservoirs
Clean up water blocking in gas reservoirs by microwave heating: Laboratory studies
Study on water locking technology of PW low permeability gas reservoir
Extracting the benefits of nanotechnology for the oil industry
Recent progress of application of nanomaterials in drilling/completion fluids and reservoir protection
The research of water lock removal for low permeability sandstone reservoirs in Chunhua Oilfield
Su 6 well gas reservoir area and reduce the harm factor analysis method of water block damage
Water removal from porous media by gas injection: Experiments and simulation
A successful methanol treatment in a gas-condensate reservoir: Field application
Evaluation of alcohol-based treatments for condensate banking removal
Selection, evaluation, and formulation of drying agents for tight sand reservoirs
Development and screening of drying agents for low permeability tight gas reservoirs
Analysis and evaluation of bound water drying reaction and drying effect in tight reservoir
Study on formula optimization and mechanism of drying agent for tight gas reservoir
Research on salting out during formation water evaporation in oil-gas reservoirs
The precipitation of salt in gas producing wells
An experimental study to evaluate water vaporization and formation damage caused by dry gas flow through porous media
Study on improvement and formulation optimization of drying agent for tight gas reservoir
Modification of the ultrafine Al4C3 powder used in the dry processing of the tight gas reservoir
Delaying the effect of an aluminum carbide drying agent in a tight gas reservoir
DOI:10.1063/1.5020111 URL [Cited within: 2]
Study on solubility and solubilization of drying agent in supercritical carbon dioxide
Study on solubility and solubilisation of drying agent in supercritical carbon dioxide for improving local permeability of tight gas reservoir
A generalized equation of state for associating fluids in nanopores: Application to CO2-H2O, CH4-H2O, CO2-CH4, and CO2-CH4-H2O systems and implication for extracting dissolved CH4 by CO2 injection
DOI:10.1016/j.ces.2020.116034 URL [Cited within: 1]
Predictions of entropy for diatomic molecules and gaseous substances
DOI:10.1016/j.cplett.2017.12.013 URL [Cited within: 1]
Phase equilibriarium modeling for confined fluids in nanopores using an association equation of state
DOI:10.1016/j.supflu.2020.105118 URL [Cited within: 1]
The drying effect and flow characteristics in porous media were quantitatively characterized by NMR online displacement
Laws of gas and water flow and mechanism of reservoir drying in tight sandstone gas reservoirs
Quantitative study of different patterns of microscale residual water and their effect on gas permeability through digital core analysis
DOI:10.1016/j.petrol.2020.108053 URL [Cited within: 1]
Flow simulation of tight gas reservoir considering reservoir drying effect
Analysis and evaluation of dryness effect of tight sandstone reservoir
Experimental study on the effect of drying on the seepage of low permeability sandstone reservoir
A basic model of unconventional gas microscale flow based on the lattice Boltzmann method
Effect of pore throat structure on micro-scale seepage characteristics of tight gas reservoirs
Simulation of a multistage fractured horizontal well in a tight oil reservoir using an embedded discrete fracture model
Simulation of a multistage fractured horizontal well in a water-bearing tight fractured gas reservoir under non-Darcy flow
DOI:10.1088/1742-2140/aaa5ce URL [Cited within: 2]