Introduction
With the surging demand for oil and gas, the number of deep-water offshore drilling wells is increasing year by year, but the drilling fluid technology is facing great challenges [1,2]. Deep-water drilling often encounters sensitive and weakly-cemented formations, resulting in easily losing wellbore stability, and generating natural gas hydrates and blocking pipelines at the low-temperature and high-pressure area near the mudline. Oil-based drilling fluid is the first choice for deep-water drilling owing to its excellent inhibition and stability. However, its rheology is greatly affected by temperature and it will thicken vigorously in deep-water low-temperature environments, resulting in a substantial increase of equivalent circulating density (ECD), consequently lost circulation and wellbore collapse in a narrow safety density window [3]. In addition, traditional base oils used in oil-based drilling fluid such as diesel oil are highly biologically toxic, so they are unable to meet the increasingly stringent marine environmental protection requirements, which greatly restrict their use in deep water drilling [4]. At the beginning of the 21st century, using environmentally friendly synthetic base oils with good viscosity-temperature characteristics combined with key treatment agents such as organic clays and rheology modifiers, the flat-rheology synthetic base drilling fluid with stable key rheological parameters such as yield point, gel strength and viscosity at low shear rates at a certain temperature range (usually 4-65 °C) was developed. It has stable ECD and environmental protection performance under the environment of large temperature difference, which realizes "safe, efficient, and environmentally friendly" deep water drilling [4]. After years of development, the formula of flat-rheology synthetic base drilling fluid has been simplified, and applicable temperature difference range has been broadened. It has been widely- used in deepwater drilling and has achieved good benefits [5,6,7]. At present, well-developed flat-rheology synthetic base drilling fluid systems at home and abroad mainly include RHELIANT PLUS from M-I SWACO, BaraECD from Baroid, NSURE from Baker Hughes, and FLAT-PRO from China Oilfield Services Co., Ltd. (COSL). The applicable temperature of FLAT-PRO reaches 3-180 °C, which surpasses the current international similar technologies [7].
However, the synthetic base oil used in flat-rheology drilling fluid is a fine chemical product with problems such as high cost, high dependence on raw petrochemical materials and non-renewability. It is more and more difficult to adapt to the low-cost requirement of oil and gas exploration and development at largely fluctuating oil prices and the demand on green and sustainable development. Therefore, in recent years, research has been carried out on preparing new biodiesel used as base oil by transesterification of animal and vegetable oils with short-chain alcohols. Li et al. [8] used kitchen waste oil to prepare biodiesel and used it as base oil to prepare a biodiesel-based drilling fluid system. It has low biotoxicity, high degradability and equivalent basic performances comparable to traditional oil-based drilling fluids, but the temperature resistance is poor. When temperature exceeds 120 °C, its viscosity will lose control due to hydrolysis, saponification and thickening of biodiesel, and the rheology is very sensitive to temperature. Dankwa et al. [9] used waste catering oil to prepare biodiesel and formulated an oil-based drilling fluid with it as the base oil. It has excellent environmental performance but it is sensitive to temperature. Its yield point and gel strength decrease sharply with temperature increasing in temperature range of 25-70 °C, which is bad for solid carrying and suspension. Sulaimon et al. and Oseh et al. [10,11] used palm oil and rapeseed oil to prepare biodiesel respectively, and used the biodiesel as base oil to prepare oil-based drilling fluid systems. The basic performances of both systems are comparable to traditional oil-based drilling fluids, but both of them seriously thicken at low temperature. Mohammed et al. [12] replaced the base oil Escaid110 in flat-rheology synthetic-based drilling fluid with palm oil biodiesel, but found that it is difficult for the drilling fluid to maintain the original flat-rheology properties, and the temperature resistance decreases. It thickens significantly after aging at 160 °C. Generally speaking, the current biodiesels have the advantages of environmental protection, independence on petrochemical raw materials, renewability, etc. The oil-based drilling fluid formulated with biodiesel has the equivalent basic performance to that of the traditional oil-based drilling fluid. However, they have shortcomings such as strong temperature sensitivity, poor low-temperature fluidity, and low temperature resistance, so they cannot be used as the base oil for flat-rheology oil-based drilling fluids. The flat-rheology of oil-based drilling fluid does not only depend on base oil, but the synergistic effect of base oil, organic clays, rheology modifiers and other key treatment agents. Treatment agents compatible with biodiesel are indispensable for the development of biodiesel-based flat-rheology drilling fluids. By now relevant targeted research has not yet been carried out.
Based on the optimization of raw materials, a cheap, environment-friendly biodiesel with good low-temperature fluidity is prepared. With the biodiesel as base oil, and the optimization and development of key treatment agents compatible with biodiesel, a biodiesel-based flat- rheology drilling fluid system is established and evaluated.
1. Biodiesel
1.1. Preparation of biodiesel
As the base oil of deep-water flat-rheology drilling fluid, biodiesel needs to have good low-temperature fluidity and suitable viscosity within a large temperature range. The low-temperature fluidity of biodiesel is mainly related to the fatty acid composition of raw oil and the molecular structure of raw alcohol [13]. The higher the saturated fatty acid content in the raw oil, the higher the condensation point of the biodiesel produced, and the easier it thickens or even solidifies at low temperature. This is because the carbon atoms on the carbon chains of the saturated fatty acid are equivalent sp3 hybrids, which are arranged in a broken line at an angle of 109.5°, making the intermolecular force of saturated fatty acid esters (van der Waals force) stronger. The double-bond carbon atoms in unsaturated fatty acid are sp2 hybridized, and the angle between the three sp2 hybrid orbitals is 120°, which makes the carbon chain of the fatty acid ester bend. The greater the number of double bonds, the greater the degree of bending, the looser the molecular arrangement, the weaker the intermolecular force and the harder it is to thicken at low temperature. The longer the carbon chain of the raw alcohol or the higher the degree of branching, the better the low-temperature fluidity of the biodiesel produced. This is because the long or highly branched carbon chain increases the distance between fatty acid ester molecules and makes it difficult for the molecules to arrange closely, resulting a weak intermolecular force. Based on the analysis, this study chose genetically-modified soybean oil with the least saturated fatty acid content and the lowest price as the raw oil to prepare biodiesel, with a saponification value of 190 mg/g, an acid value of 1.25 mg/g, and an average molecular weight of 890. The fatty acid composition of genetically modified soybean oil was analyzed by a gas chromatography-mass spectrometry. The results show that more than 80% of the composition is unsaturated acids with 18 carbon atoms (Table 1).
Table 1 Fatty acid composition of genetically-modified soybean oil
| Composition | Content/% | Carbon atom number | Unsaturated bond number |
|---|---|---|---|
| Octoic acid | 0.02-0.03 | 8 | 0 |
| Tetradecanoic acid | 0.10-0.20 | 14 | 0 |
| Pentadecanoic acid | 0.02-0.03 | 15 | 0 |
| 9-Hexadecenoic acid | 0.10-0.20 | 16 | 1 |
| Palmitic acid | 1.50-1.60 | 16 | 0 |
| Heptadecanoic acid | 0.20-0.30 | 17 | 0 |
| Linoleic acid | 51.00-52.00 | 18 | 2 |
| Oleic acid | 30.00-31.00 | 18 | 1 |
| Stearic acid | 8.00-9.00 | 18 | 0 |
| Docosanoic acid | 1.00-2.00 | 22 | 0 |
In order to optimize the raw alcohol, the genetically- modified soybean oil is transesterified with different alcohols to prepare biodiesel. Specific steps: (1) Weigh the genetically-modified soybean oil and the alcohol according to a certain molar ratio (simplified as the alcohol-oil ratio), and preheat the genetically-modified soybean oil for 1 h at a predetermined reaction temperature; (2) Add a certain amount of catalyst to the alcohol and stir at the reaction temperature until it dissolves; (3) Mix the catalyst-alcohol solution with the preheated genetically-modified soybean oil and stir at the reaction temperature at the constant temperature until the reaction is over, then let the mixture stand for 24 h and use a separatory funnel to separate the supernatant to obtain crude biodiesel product; (4) Wash the crude product 3 times with water and place it in a rotary evaporator to distill the remaining alcohol and obtain a final biodiesel product. In this paper, referring to the method of Xu et al. [14], the yield of biodiesel is obtained by dividing the mass of biodiesel by the mass of the raw oil used.
The experimental results show that the reaction conditions of methanol and genetically-modified soybean oil are the mildest, with the highest reaction rate and yield. The alcohol-oil ratio and reaction temperature of ethanol and genetically-modified soybean oil are slightly higher than methanol with genetically-modified soybean oil, the reaction rate is slightly slower than that of methanol, and the yield can reach more than 90%. Compared with the former two, the alcohol-oil ratio and reaction temperature required for the reaction of both propanol and isopropanol with genetically-modified soybean oil are greatly increased, the reaction rate is slow, and the final yield is low (Table 2). In general, the longer the alcohol molecular chain, the more complex the structure, the greater the steric hindrance, the higher the conditions required for the transesterification reaction, and the lower the degree of reaction. At present, among the base oil commonly-used in oil-based drilling fluids, 0# diesel oil is about 4000 RMB/t, white oil is about 5500 RMB/t, synthetic base oil like Escaid110, gas-made oil and polylinear α-olefins is about 15 000-20 000 RMB/t, the cost of biodiesel made from methanol and ethanol is lower than 0# diesel and white oil, so it has better price advantages compared with synthetic base oil. The cost of biodiesels made from propanol and isopropanol are high and have no advantages (Table 2).
Table 2 Optimal reaction condition, yield and cost of biodiesel prepared with different raw alcohols
| Alcohol | Alcohol-oil ratio | Catalyst | Catalyst addition/% | Temperature/°C | Time/h | Yield/% | Cost/(RMB·t-1) |
|---|---|---|---|---|---|---|---|
| Methanol | 5:1 | NaOH | 1 | 50 | 0.5 | 96 | 3400 |
| Ethanol | 6:1 | NaOH | 1 | 65 | 1.0 | 92 | 5200 |
| Propanol | 10:1 | NaOH | 2 | 90 | 2.0 | 44 | 15 000 |
| Isopropanol | 12:1 | Enriched H2SO4 | 5 | 80 | 5.0 | 27 | 17 000 |
1.2. Regular properties of biodiesel
In this study, the regular properties of self-made biodiesel were tested and compared with other commonly- used base oils (Table 3). The closed flash points of self- made biodiesel are much higher than that of commonly- used base oils and are safer. With the increase of the carbon chain length and branching degree of the raw alcohol, the viscosity of biodiesel gradually increases and the pour point gradually decreases. The pour points of the biodiesels made from methanol and ethanol are -1 °C and -5 °C respectively, which can meet the requirements of deepwater environment. Although the biodiesel prepared from propanol and isopropanol has a lower pour point, the viscosity is too high for the rheology of drilling fluid. Although methanol has the lowest price, it has strong biological toxicity. Ethanol has low biological toxicity and a broad resource of raw materials, and more environment-friendly and reproducible. Based on the considerations above, the soybean oil ethyl ester biodiesel prepared from genetically-modified soybean oil and ethanol is selected as the final base oil for flat-rheology drilling fluid.
Table 3 Regular properties of different oils
| Oil | Appearance | Density (20 °C)/ (kg·m-3) | Kinematic viscosity (40 °C)/(mm2·s-1) | Closed flash point/°C | Pour point/°C |
|---|---|---|---|---|---|
| Methanol-made biodiesel | Light yellow fluid | 891.6 | 3.8 | 133 | -1 |
| Ethanol-made biodiesel | Light yellow fluid | 882.4 | 4.1 | 142 | -5 |
| Propanol-made biodiesel | Light yellow fluid | 874.1 | 6.2 | 146 | -11 |
| Isopropanol-made biodiesel | Light yellow fluid | 872.9 | 6.8 | 165 | -16 |
| Escaid110 | Transparent fluid | 790.0 | 1.7 | 70 | -30 |
| Saraline185V | Transparent fluid | 770.0 | 2.8 | 82 | -20 |
| 3# white oil | Transparent fluid | 800.0 | 3.2 | 80 | -3 |
| 5#white oil | Transparent to light yellow fluid | 820.0 | 5.0 | 110 | -5 |
| 0# diesel | Light yellow fluid | 850.0 | 3.5 | 55 | -18 |
1.3. Environmental protection of biodiesel
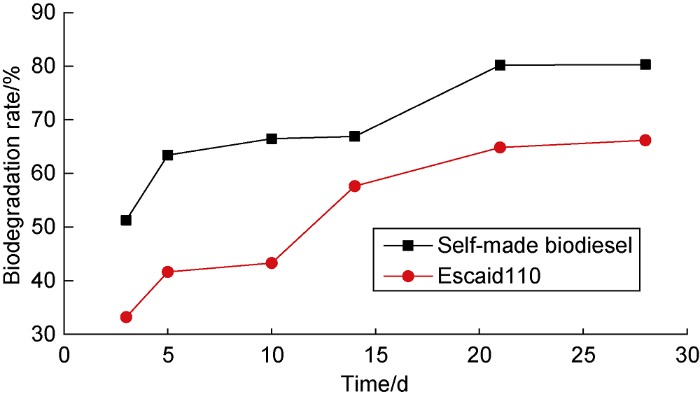
The biodegradability and biotoxicity of the soybean oil ethyl ester biodiesel are evaluated by referring to "Chemicals-biodegradability in Seawater-closed Bottle Method"[15] and "Biological Toxicity for Pollutants from Marine Petroleum Exploration and Exploitation-Part 2: Test Method" [16], and compared with Escaid110 which has excellent environmental protection among the commonly-used synthetic base oils (Fig. 1, Table 4). The self-made biodiesel has a higher biodegradation rate than Escaid110, the final degradation rates of the two in 28 d are 80.27% and 66.16%, respectively. The self-made biodiesel has a more than 1 000 000 mg/L of median lethal concentration (LC50) in 96 h for Artemia, which is almost completely non-toxic. The 96 h LC50 value of Escaid110 for Artemia is 39 765.7-80 585.3 mg/L. Although it is higher than the allowable limit (15 000 mg/L) specified for the first-level sea area, it still has obvious biological toxicity. Compared with traditional synthetic base oils, the self-made soybean oil ethyl ester biodiesel has better environmental performance.
Fig. 1.
Fig. 1.
Biodegradation rates of self-made biodiesel and Escaid110.
Table 4 Biological toxicity of self-made biodiesel and Escaid110
| Oil | Sample | Number of Artemia | Dead Artemias (96 h) | Artemia death rate (96 h)/% | 96 h LC50/ (mg·L-1) |
|---|---|---|---|---|---|
| Self-made biodiesel | Blank | 40 | 0 | 0 | >1 000 000.0 |
| Saturated aqueous solution | 40 | 0 | 0 | ||
| Escaid110 | Blank | 40 | 0 | 0 | 39 765.7-80 585.3 |
| 50 times diluted solution | 40 | 4 | 10 |
2. Key treatment agents
2.1. Formula optimization of biodiesel-based emulsion
2.1.1. The best range of hydrophilic-lipophilic balance value
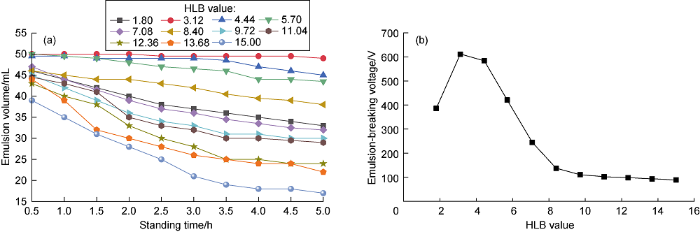
To determine the most suitable hydrophile-lipophile balance (HLB) value of oil phase and water phase is the key to selecting oil-based drilling fluid emulsifiers. Taking Span85 with an HLB value of 1.8 and Tween80 with an HLB value of 15.0 as emulsifiers, a combination of emulsifiers with different HLB values is obtained by compounding them in different proportions. The combined HLB value is equal to the addition of the product of HLB value and mass fraction of each emulsifier. Using self- made biodiesel as oil phase and CaCl2 brine (25% mass fraction) as water phase (the same below), the oil-water ratio is set to be 90 to 10, and the emulsifier addition is set to be 3% of the total volume of oil and water. Emulsifiers with different HLB values are used to formulate different emulsions. The amount of each component in the drilling fluid formula in this paper is calculated using this method. Pour each emulsion into a 50 mL graduated cylinder, let it stand at room temperature and record the change in its volume over time to evaluate the stability of settlement; at the same time, test the breaking voltage of the emulsion at 65 °C (the same below). It can be seen from Fig. 2a that the biodiesel-based emulsion prepared with a combination of emulsifiers with HLB values of 3.12, 4.44, and 5.70 has a large volume after standing for 5 h, indicating that the oil-water separation is weak and the settlement stability is good, which is consistent with the evaluation results of the electrical stability (Fig. 2b). The best range of the emulsifier HLB value is 3-6 for self-made biodiesel emulsion.
Fig. 2.
Fig. 2.
The influence of HLB value of compound emulsifier on sendimentation stability (a) and emulsion-breaking voltage (b) of biodiesel-based emulsion.
2.1.2. Optimization of oil-water ratio
According to the optimal range of HLB value, Span80 with the HLB value of 4.3 is selected as the only emulsifier, and biodiesel-based emulsions with different oil-water volume ratios (oil-water ratio in brief) are prepared according to the formulas in Table 5. The oil-water ratio is optimized.
Table 5 Formulas of biodiesel-based emulsions with different oil-to-water ratios
| Oil-water ratio | Self-made biodiesel/mL | Span80/g | CaCl2 brine/mL | CaO/g |
|---|---|---|---|---|
| 90:10 | 270 | 9 | 30 | 3 |
| 80:20 | 240 | 9 | 60 | 3 |
| 70:30 | 210 | 9 | 90 | 3 |
where τ is shear stress, Pa; τy is yield point, Pa; K is consistency coefficient, Pa•sn; γ is shear rate, s-1; n is fluidity index, dimensionless.
Fig. 3.
Fig. 3.
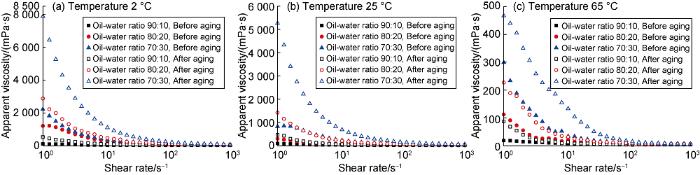
Influence of oil-water ratio on the apparent viscosity of biodiesel-based emuslion before and after aging at different temperature.
Fig. 4.
Fig. 4.
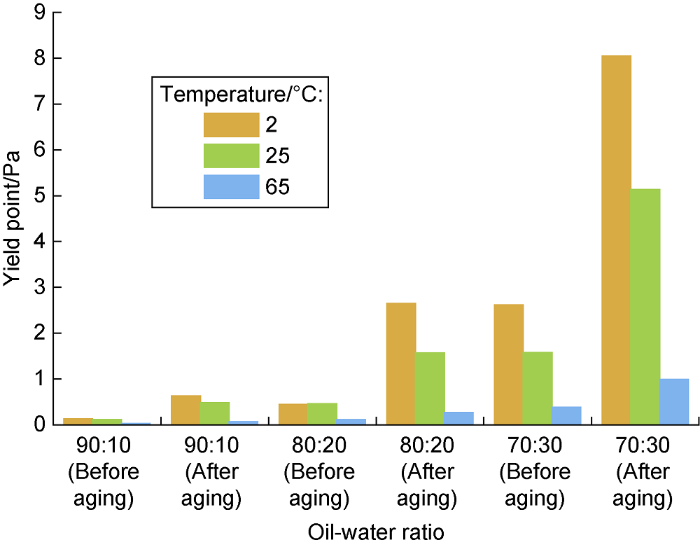
Influence of oil-water ratio on the yield point of biodiesel-based emuslion before and after aging at different temperatures.
The experimental results show that after aging at high temperature, the viscosity and strength of the emulsion increase, and the emulsion with higher water content behaves more significant. After aging at high temperature, the low-temperature thickening phenomenon of the emulsion become more severe, and the higher the water content, the more significant it is. This comes from the hydrolysis of fatty acid esters at high temperatures, and the hydrolysate reacts with Ca2+ in the emulsion and forms calcium soap [17]. Because the thickening caused by hydrolysis reaction is more severe at low temperature, which is not conducive to controlling the rheology of drilling fluid in deep water environment. A higher oil-to- water ratio is benefit for constructing a flat-rheology biodiesel-based drilling fluid system, so the final ratio is 90:10.
2.1.3. Optimal combination of emulsifiers
The fixed oil-water ratio is 90:10. 1% primary emulsifier (Span80), 1% combination of secondary emulsifiers and 1% CaO are added to prepare a biodiesel-based emulsion with different combinations of emulsifiers. Emulsion 1 is made up of nonionic surfactant Span85 and anionic sodium dodecylbenzene sulfonate with an HLB value of 10.6, with mass ratio of 6:4. Emulsion 2 is made up of Span85 and cationic octadecyltrimethyl ammonium chloride with an HLB value of 15.7, with mass ratio of 7:3. Emulsion 3 is made up of Span85 and non-ionic Tween80 with an HLB value of 15.0, with mass ratio of 7:3.
The breaking voltages of the emulsions before and after aging at 150 °C for 16 h were tested respectively (Table 6). The breaking voltages of the emulsions decrease after aging at high temperature, but they are all higher than 400 V, which meets the electrical stability of oil-based drilling fluids.
Table 6 Breaking voltages of biodiesel-based emulsions before and after aging
| Sample | Condition | Breaking voltage/V |
|---|---|---|
| Emulsion 1 | Before aging | 686 |
| After aging | 458 | |
| Emulsion 2 | Before aging | 615 |
| After aging | 583 | |
| Emulsion 3 | Before aging | 702 |
| After aging | 549 |
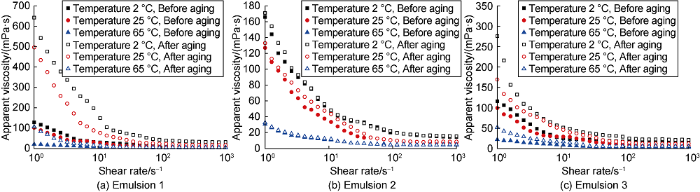
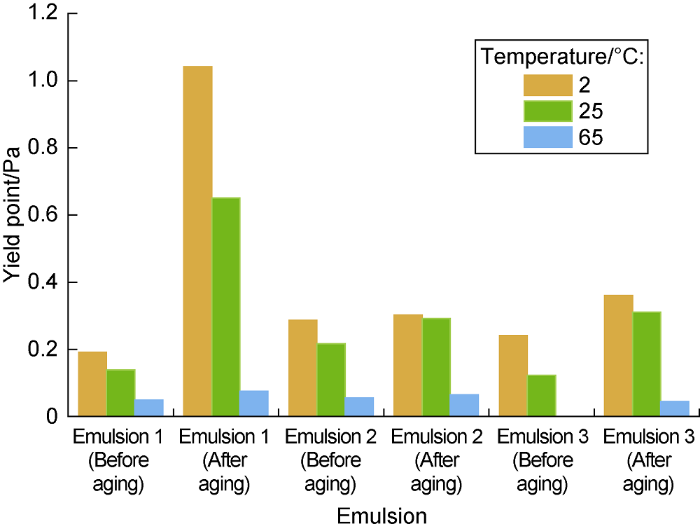
The viscosity of the emulsion before and after aging tested at different temperatures (Fig. 5) and the yield point fitted by Herschel-Bulkley model (Fig. 6) show that, when the secondary emulsifier contains anionic surfactant (Emulsion 1), the viscosity at low shear rates and yield point of the emulsion increase significantly after high temperature aging, and the low-temperature thickening phenomenon is obvious. When the secondary emulsifier contains cationic surfactant (Emulsion 2), the viscosity at low shear rates and yield point of the emulsion before and after aging have the smallest changes tested at different temperature. When the secondary emulsifier is a composite nonionic surfactant (Emulsion 3), the emulsion also exhibits a certain increase of viscosity after aging with the amplitude smaller than that of Emulsion 1. It indicates that cationic surfactants have the best inhibitory effect on the hydrolysis of fatty acid esters under alkaline conditions, thereby effectively preventing the saponification and thickening of biodiesel-based emulsions from high-temperature aging. Finally, the optimal emulsifier system is composed of Span80 as the primary emulsifier and secondary combination II (Span85 + octadecyltrimethyl ammonium chloride).
Fig. 5.
Fig. 5.
Viscosity curves of biodiesel-based emulsion before and after aging at different test temperature.
Fig. 6.
Fig. 6.
Yield points of biodiesel-based emulsions before and after aging at different test temperature.
2.2. Optimization of organic clay
In the previous study, a series of quaternary ammonium salt cationic surfactants with different lengths and numbers of carbon chain were used to modify sodium- based montmorillonite through cation exchange reaction to prepare a series of organic clays (Table 7) [18]. By studying the relationship between the dispersibility and rheology of various organic clays in soybean oil ethyl ester biodiesel at 2-65 °C, it is found that the closer the polarity of the cationic modifier and the biodiesel, the higher the affinity between the organic clay and the biodiesel, the easier the organic clay disperses stably, and the less the rheology of the resulting dispersion affected by temperature. The dispersion stability of the organic clays in biodiesel is OC16, OC18, ODC16, OTC16, ranked from strong to weak in turn.
Table 7 Basic performance of self-made organic clays
| Organic clay | Cationic modifier | Interlayer spacing/ nm | Average particle size/μm | Colloidal rate in biodiesel/% |
|---|---|---|---|---|
| OC16 | Cetyltrimethyl ammonium chloride | 1.979 | 3.58 | 88 |
| OC18 | Octadecyltrimethyl ammonium chloride | 1.998 | 5.13 | 72 |
| ODC16 | Double-cetyltrimethyl ammonium chloride | 3.635 | 6.15 | 54 |
| OTC16 | Triple-cetyltrimethyl ammonium chloride | 4.492 | 7.07 | 46 |
270 mL self-made biodiesel, 1% Span80, 1% secondary emulsifier combination II, 30 mL of CaCl2 brine, 1% CaO and barite were mixed to prepare a biodiesel-based drilling fluid with a density of 1.2 g/cm3. After adding 2% of each type of organic clay to the above drilling fluid respectively, and aging at 150 °C for 16 h, tested the rheology (by a GDC-II rheometer), emulsion-breaking voltage and HPHT filtrate of the drilling fluid at 2-65 °C (Table 8). The test results show that the rheology of the drilling fluid with organic clay OC16 changes the least with temperature, the emulsion-breaking voltage is more than 2000 V, the stability is strong, and the filtrate is the lowest. As the length and quantity of the carbon chains of the cationic modifier in the organic clay increase, the low-temperature thickening of the drilling fluid gradually becomes intense, the breaking voltage gradually decreases, and the filtrate gradually increases. Finally, organic clay OC16 is selected.
Table 8 Influence of organic clay on performance of biodiesel-based drilling fluid
| Organic clay | Temperature/ °C | Apparent viscosity/ (mPa·s) | Plastic viscosity/ (mPa·s) | Yield point/ Pa | Φ6 | Φ3 | Initial gel strength/ Pa | Final gel strength/ Pa | Emulsion- breaking voltage/V | HPHT filtrate/mL |
|---|---|---|---|---|---|---|---|---|---|---|
| OC16 | 2 | 43.0 | 38 | 5.11 | 5 | 4 | 2.0 | 2.5 | 2 035 | 18.1 |
| 30 | 25.0 | 21 | 4.09 | 4 | 3 | 1.5 | 2.0 | |||
| 65 | 14.0 | 11 | 3.07 | 3 | 2 | 1.0 | 2.0 | |||
| OC18 | 2 | 46.0 | 40 | 6.13 | 6 | 5 | 2.5 | 3.0 | 1 653 | 22.2 |
| 30 | 27.0 | 23 | 4.09 | 3 | 2 | 1.0 | 1.5 | |||
| 65 | 12.0 | 10 | 2.04 | 2 | 2 | 1.0 | 1.0 | |||
| ODC16 | 2 | 47.0 | 40 | 7.15 | 7 | 6 | 3.0 | 3.5 | 1 132 | 29.1 |
| 30 | 31.0 | 26 | 5.11 | 4 | 3 | 1.5 | 2.0 | |||
| 65 | 15.5 | 13 | 2.56 | 3 | 2 | 1.0 | 1.5 | |||
| OTC16 | 2 | 52.5 | 43 | 9.71 | 8 | 7 | 3.0 | 3.5 | 791 | 32.0 |
| 30 | 33.0 | 27 | 6.13 | 5 | 4 | 2.0 | 2.5 | |||
| 65 | 16.0 | 13 | 3.07 | 3 | 2 | 1.0 | 1.5 |
Note: The rheological parameters of the drilling fluid are calculated based on the Bingham model; Φ6, Φ3 are readings on a rheometer with speed at 6, 3 r/min respectively; the temperature for measuring HTHP filtrate is identical to the aging temperature; the pressure difference is 3.5 MPa.
2.3. Flat-rheology modifier
2.3.1. Synthesis and characterization
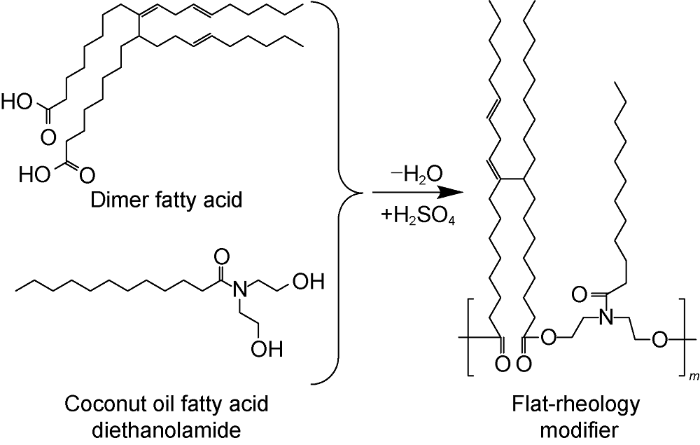
The main components of the flat-rheology modifier (PER) are dimer fatty acid and coconut oil fatty acid diethanolamide which are condensed at a molar ratio of 1:1. It is essentially an amphiphilic polymer synthesized following the route shown in Fig. 7. Specific synthesis procedures: weigh 56 g dimer fatty acid and 29 g coconut oil fatty acid diethanolamide into a 3-neck flask; add 0.1 g catalyst (concentrated H2SO4), and protected by nitrogen and at 250 r/min, increase to 100 °C and keep for 30 min; then quickly increase to 200 °C, and keep reacting until no water is produced. Finally, the number average relative molecular weight of the PER is 7600 detected by a gel permeation chromatography analyze.
Fig. 7.
Fig. 7.
Synthesis route of flat-rheology modifier (m is polymerization degree).
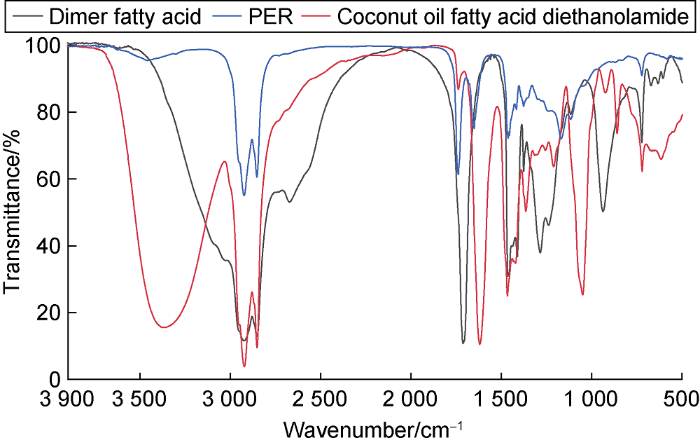
The infrared spectra of dimer fatty acid, coconut oil fatty acid diethanolamide and PER show (Fig. 8) the absorption peak of dimer fatty acid at 2923 cm-1 and 2852 cm-1 is stretching vibration peak of C-H in alkyl, and stretching vibration peak of C=O in carboxyl at 1712 cm-1; coconut oil fatty acid diethanolamide has a stretching vibration peak of O-H in hydroxyl at 3359 cm-1, stretching vibration peaks of C-H in alkyl at 2923 cm-1 and 2850 cm-1, and the stretching vibration peak of C=O in the amide group at 1619 cm-1; in the PER spectra, the characteristic peaks of hydroxyl and carboxyl groups disappear, and the characteristic peak of amide group at 1619 cm-1 of coconut oil fatty acid diethanolamide is retained. At the same time, an obvious ester group C=O characteristic peak appears at 1740 cm-1, which proves the successful esterification reaction between dimer fatty acid and coconut diethanolamide.
Fig. 8.
Fig. 8.
The infrared spectra of dimer fatty acid, coconut oil fatty acid diethanolamide and PER.
2.3.2. Optimal PER addition
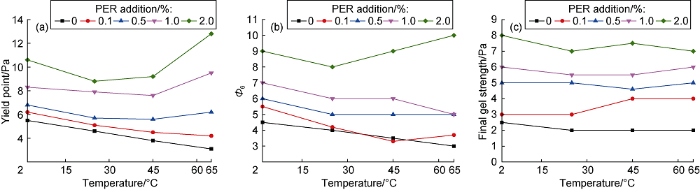
Add different amounts of PER to the biodiesel-based drilling fluid prepared above, and measure the change of the three key rheological parameters after aging for 16 h at 160 °C, namely, the yield point, reading on the rheometer at 6 r/min (Φ6) and final gel strength (Fig. 9). It is found that with the increase of PER, the three rheological parameters gradually increase at any test temperature. When adding 0.5% PER, the rheological parameters remain almost constant at 2-65 °C, showing excellent flat-rheology. Finally, the optimal addition of PER is determined to be 0.5%.
Fig. 9.
Fig. 9.
Influence of PER addition on the yield point (a), Φ6 (b) and final gel strength of the biodiesel-based drilling fluid system.
2.3.3. Mechanism
A Leica DM4M optical microscope was used to observe the effect of additional PER on the microscopic morphology of the biodiesel-based emulsion (Fig. 10). The emulsion droplets without PER are dispersed uniformly. After increasing PER, the emulsion droplets begin to gather and connect to a spatial network. This is because the amphiphilic molecular structure of the PER allows it to be adsorbed on the interfacial films of multiple emulsion droplets at the same time, thereby creating bridging connections among the emulsion droplets, forming a gel-like network, and effectively improving the viscosity and strength of the emulsion. Thickening and strengthening effect of PER gradually become stronger as temperature rises, because its molecular chain gradually stretches with the increase of temperature, so that it can be absorbed and bridged with more emulsion droplets to form a denser and stronger network [19]. Although adding more PER can create a stronger structure, it is not conductive to the flat-rheology performance of the drilling fluid when adding PER more than 0.5% according to the optimization results. In conclusion, it is no need to add too much PER.
Fig. 10.
Fig. 10.
Influence of PER addition on the microscopic morphology of biodiesel-based emulsion. (a) The emulsion droplets are uniformly dispersed without adding PER; (b) Some emulsion droplets gather together when adding 0.1% PER; (c) The emulsion droplets gather and connect with each other when adding 0.5% PER; (d) Many emulsion droplets gather and connect to a gel network when adding 1.0% PER; (e) Many emulsion droplets gather and form a denser gel network when adding 2.0% PER.
3. Formulation and performance evaluation of biodiesel-based flat-rheology drilling fluid system
Considering that the primary component of biodiesel is unsaturated fatty acid ester which has poor oxidation stability, tertbutyl hydroquinone (TBHQ) is used as an antioxidant. In order to further enhance the hydrolytic stability of the drilling fluid, carbodiimide (AW700) is added as a hydrolysis stabilizer. Its C=N bond is more active than the ester bond in biodiesel, and the hydrolysis reaction takes place more preferentially, so that the hydrolysis of biodiesel can be avoided to a large extent. The final formula of the biodiesel-based flat-rheology drilling fluid system includes 270 mL of self-made biodiesel, 30 mL of CaCl2 brine, 1% Span80, 1% secondary emulsifier combination II, 1% CaO, 0.5% PER, 2% OC16, 0.5% TBHQ, 2% AW700, 1% oxidized pitch, 3% CaCO3. The drilling fluid is weighted with barite to different densities.
3.1. Basic performance
The rheology, emulsion-breaking voltage and API filtrate of the biodiesel-based flat-rheology drilling fluid tested at 2-65 °C after aging for 16 h at 160 °C show that (Table 9) the emulsion-breaking voltage is more than 1500 V, indicating an excellent stability; when the density is 1.2 g/cm3, the change rate of yield point, Φ6, Φ3, initial gel strength and final gel strength with temperature are all less than 20%, indicating that the drilling fluid has excellent flat-rheological properties; when the density is 1.5 g/cm3, it still has flat-rheological property, but as the density continues to increase, the viscosity and yield point increase significantly, the low-temperature thickening is intensified and the flat-rheological property is gradually lost.
Table 9 Basic performance of biodiesel-based flat-rheology drilling fluid
| Density/ (g·cm-3) | Test temperature/°C | Apparent viscosity / (mPa·s) | Plastic viscosity / (mPa·s) | Yield point/ Pa | Φ6 | Φ3 | Initial gel strength/ Pa | Final gel strength/ Pa | Emulsion- breaking voltage/V | API filtrate/ mL | HTHP filtrate/mL |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1.2 | 2 | 48 | 39 | 9.20 | 8 | 7 | 3.5 | 4 | 2 035 | 1.4 | 8.2 |
| 30 | 35 | 27 | 8.18 | 7 | 6 | 3.0 | 4 | ||||
| 65 | 18 | 10 | 8.18 | 7 | 6 | 3.0 | 4 | ||||
| 1.5 | 2 | 55 | 44 | 11.24 | 12 | 11 | 5.5 | 6 | 2 021 | 0.8 | 6.4 |
| 30 | 39 | 30 | 9.20 | 10 | 9 | 4.5 | 5 | ||||
| 65 | 28 | 20 | 8.18 | 9 | 8 | 4.0 | 5 | ||||
| 1.8 | 2 | 64 | 47 | 17.37 | 15 | 13 | 6.5 | 8 | 1 656 | 2.8 | 11.4 |
| 30 | 54 | 42 | 12.26 | 12 | 11 | 5.5 | 6 | ||||
| 65 | 47 | 36 | 11.24 | 10 | 9 | 6.0 | 8 | ||||
| 2.0 | 2 | 76 | 53 | 23.51 | 88 | 20 | 10.0 | 11 | 1 597 | 3.2 | 12.8 |
| 30 | 65 | 51 | 14.31 | 19 | 17 | 9.0 | 10 | ||||
| 65 | 56 | 47 | 9.20 | 14 | 13 | 7.0 | 8 |
The Fann-iX77 rheometer (high-temperature and high- pressure rheometer) was used to test the flat-rheology performance of the biodiesel-based flat-rheology drilling fluid with a density of 1.2 g/cm3 in a wider temperature range. The results show (Table 10) that the upper limit of temperature at which the drilling fluid can maintain flat-rheology is 90 °C, and the rheological parameters are greatly reduced after temperature continues to rise.
Table 10. HTHP rheology of the biodiesel-based flat-rheology drilling fluid with a density of 1.2 g/cm3
| Temperature/°C | Pressure/MPa | Apparent viscosity/(mPa·s) | Plastic viscosity/(mPa·s) | Yield point/Pa | Φ6 | Φ3 |
|---|---|---|---|---|---|---|
| 70 | 34.29 | 36.7 | 27.5 | 9.40 | 10.8 | 9.6 |
| 90 | 44.59 | 29.5 | 20.9 | 8.79 | 9.3 | 7.7 |
| 120 | 58.78 | 24.8 | 18.5 | 6.44 | 6.6 | 5.4 |
| 150 | 73.48 | 19.3 | 16.2 | 3.17 | 3.7 | 2.6 |
| 180 | 88.18 | 16.5 | 15.8 | 0.71 | 1.4 | 1.1 |
| 210 | 102.87 | 16.3 | 16.1 | 0.20 | 1.2 | 0.6 |
3.2. Evaluation of anti-pollution performance
Artificial seawater and shale cuttings were added to the biodiesel-based flat-rheology drilling fluid with a density of 1.2 g/cm3, and the performance parameters were tested after aging for 16 h at 160 °C to evaluate the anti-pollution performance (Table 11). Drilling fluid is sensitive to seawater pollution. As seawater pollution increases, the viscosity and yield point increase greatly, the low- temperature thickening phenomenon intensifies, the emulsion-breaking voltage decreases and the filtrate increases. The oil-water ratio is reduced after the intrusion of seawater, which leads to poor stability of emulsification and promotes the hydrolysis and saponification of biodiesel, and consequently the out-of-control rheology of the system. When seawater pollution is less than 5%, the key flat-rheological parameters of the drilling fluid can be kept constant, while having good emulsification stability and low fluid loss. This indicates that the drilling fluid system can resist 5% of seawater pollution. In the same way, the drilling fluid system can resist 10% of shale cuttings pollution (Table 12).
Table 11. Anti-seawater performance of biodiesel-based flat-rheology drilling fluid
| Seawater addition/ % | Rheology test temperature/°C | Apparent viscosity/ (mPa·s) | Plastic viscosity/ (mPa·s) | Yield point/ Pa | Φ6 | Φ3 | Initial gel strength/ Pa | Final gel strength/ Pa | Emulsion- breaking voltage/V | API filtrate/ mL | HTHP filtrate/ mL |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 5 | 2 | 53 | 43 | 16.86 | 10 | 9 | 4.5 | 5.0 | 1 565 | 1.8 | 8.1 |
| 30 | 44 | 26 | 4.09 | 8 | 7 | 3.5 | 4.5 | ||||
| 65 | 23 | 15 | 3.58 | 8 | 7 | 3.5 | 4.0 | ||||
| 10 | 2 | 64 | 52 | 20.44 | 14 | 13 | 6.0 | 7.0 | 974 | 2.2 | 10.3 |
| 30 | 49 | 40 | 15.84 | 19 | 17 | 5.0 | 6.0 | ||||
| 65 | 34 | 27 | 10.22 | 14 | 13 | 6.0 | 6.0 |
Table 12. Anti-shale cuttings performance of biodiesel-based flat-rheology drilling fluid
| Shale cuttings addition/% | Rheology test temperature/°C | Apparent viscosity/ (mPa·s) | Plastic viscosity/ (mPa·s) | Yield point/ Pa | Φ6 | Φ3 | Initial gel strength/ Pa | Final gel strength/ Pa | Emulsion- breaking voltage/V | API filtrate/ mL | HTHP filtrate/ mL |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 5 | 2 | 52 | 44 | 8.18 | 7 | 6 | 3.0 | 4.0 | 1 353 | 0.8 | 8.1 |
| 30 | 43 | 36 | 7.15 | 7 | 6 | 3.0 | 3.5 | ||||
| 65 | 31 | 24 | 7.15 | 6 | 5 | 2.5 | 3.0 | ||||
| 10 | 2 | 58 | 49 | 9.20 | 9 | 8 | 4.0 | 5.0 | 1 138 | 1.8 | 11.4 |
| 30 | 46 | 38 | 8.18 | 8 | 7 | 3.5 | 4.0 | ||||
| 65 | 37 | 30 | 7.15 | 7 | 6 | 3.0 | 3.5 | ||||
| 15 | 2 | 64 | 50 | 14.31 | 13 | 12 | 6.0 | 7.0 | 875 | 3.6 | 16.0 |
| 30 | 52 | 41 | 11.24 | 10 | 9 | 4.5 | 5.0 | ||||
| 65 | 43 | 35 | 8.18 | 8 | 7 | 3.5 | 4.0 |
3.3. Biological toxicity
Referring to the "Biological Toxicity for Pollutants from Marine Petroleum Exploration and Exploitation-Part 2: Test Method" [16] to test the biological toxicity of the biodiesel-based flat-rheology drilling fluid, the 96 h LC50 values for moina mongolica I-year larvae and Artemia nauplii are 46 000 mg/L and 44 000 mg/L respectively, meeting the requirement of the first-level sea area (more than 30 000 mg/L), indicating excellent performance on environmental protection.
4. Conclusions
Soybean oil ethyl ester biodiesel prepared with genetically-modified soybean oil and ethanol as raw materials has good regular physical and chemical properties and low-temperature fluidity, and is more environment-friendly and lower in cost than traditional base oil used in synthetic-based drilling fluid.
The oil-water ratio, emulsifier combination and organic clay suitable for self-made biodiesel are optimized. The flat-rheology modifier synthesized with dimer fatty acid and coconut oil fatty acid diethanolamide is prepared, which can form a strong network structure with emulsion droplets, and can effectively alleviate the phenomenon that the key rheological parameters of biodiesel-based drilling fluid greatly decrease with the increase of temperature, so to keep good flat-rheological property.
A biodiesel-based flat-rheology drilling fluid system is formulated with a density of 1.2 g/cm3. It can maintain flat-rheological property at an oil-water ratio of 90:10 and a temperature range of 2-90 °C. It can resist temperature up to 160 °C, 5% seawater and 10% shale cuttings. The environmental performance is excellent.
Reference
The top 10 mud-related concerns in deepwater drilling operations
Technological progress and development directions of PetroChina overseas oil and gas field production
Research progress and prospect of plugging technologies for fractured formation with severe lost circulation
Study on gelation and freezing phenomena of synthetic drilling fluids in ultra deep water environments
Research and application of FLAT-PRO synthetic based drilling fluid system in deep water
Status quo of the development of deep water drilling fluids worldwide
China’s independent research and development of deepwater drilling fluid technology achieves “leading”
An investigation on environmentally friendly biodiesel-based invert emulsion drilling fluid
DOI:10.1007/s13202-015-0205-7 URL [Cited within: 1]
Investigating the potential use of waste vegetable oils to produce synthetic base fluids for drilling mud formulation
Performance enhancement of selected vegetable oil as base fluid for drilling HPHT formation
DOI:10.1016/j.petrol.2017.02.006 URL [Cited within: 1]
Investigating almond seed oil as potential biodiesel-based drilling mud
DOI:10.1016/j.petrol.2019.106201 URL [Cited within: 1]
The use of palm oil fatty acid methyl ester as a base fluid for a flat rheology high-performance drilling fluid
DOI:10.1016/j.petrol.2018.03.101 URL [Cited within: 1]
Study on the preparation of biodiesel and the improvement of its low temperature fluidity
Study on synthesis of soybean oil isopropyl ester
Investigation of biodiesel-based drilling fluid, part 1: Biodiesel evaluation, invert-emulsion properties, and development of a novel emulsifier package
DOI:10.2118/180918-PA URL [Cited within: 1]
Study on morphology and rheological property of organoclay dispersions in soybean oil fatty acid ethyl ester over a wide temperature range
DOI:10.1021/acsomega.9b03183 URL [Cited within: 1]
Synthetic-based drilling fluid with constant-rheology used in deepwater drilling