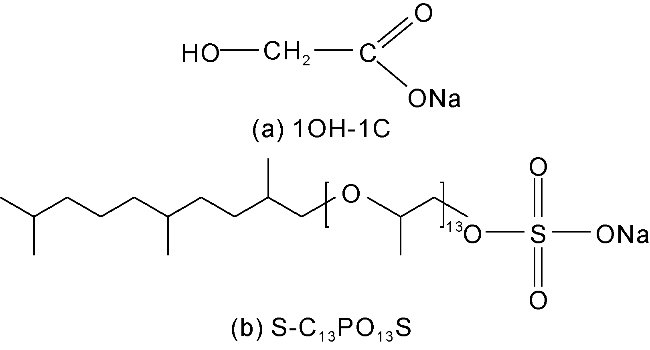
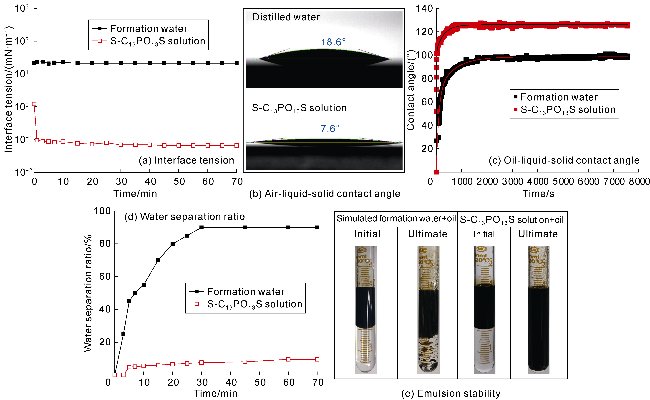
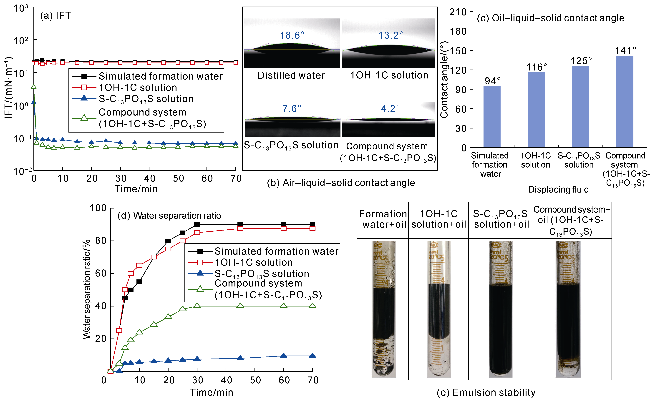
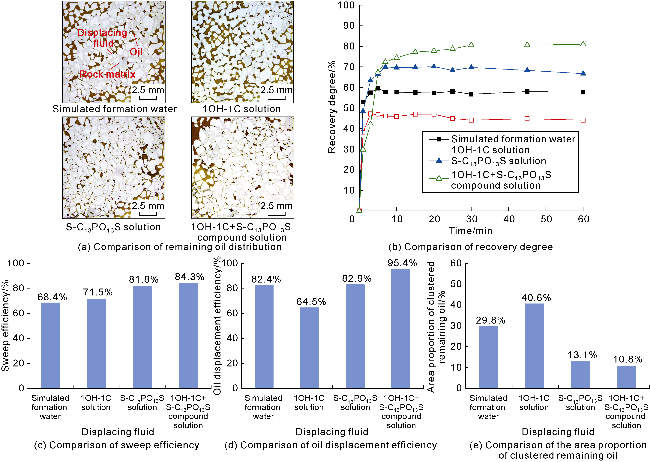
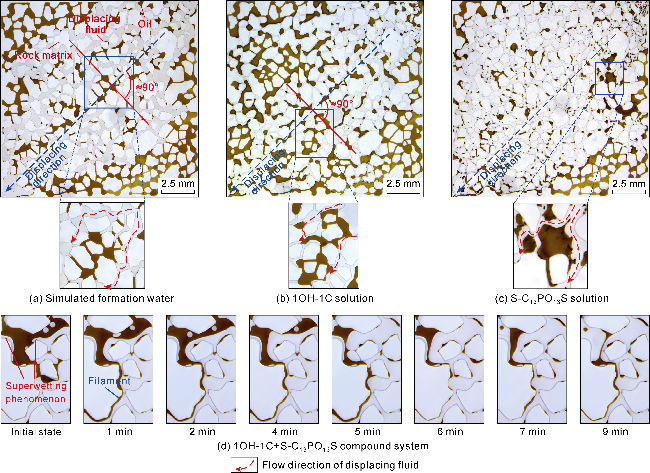
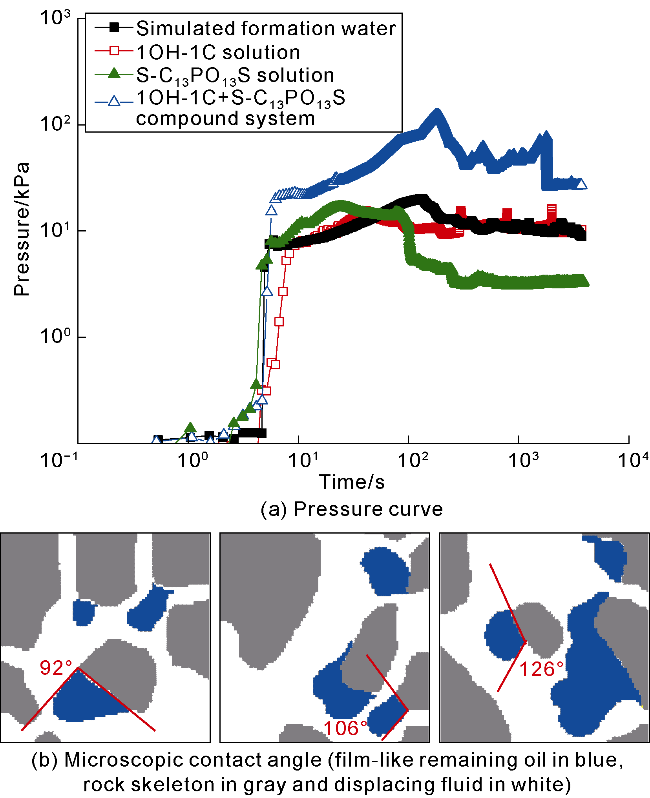
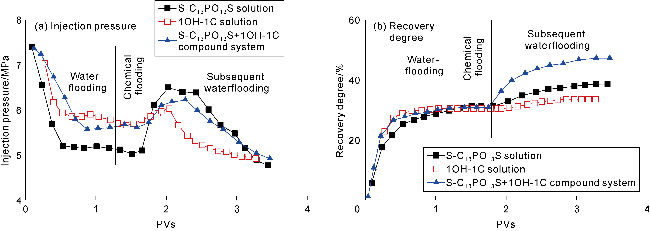
The interfacial properties of S-C
13PO
13S solution are shown in
Fig. 8. The dynamic IFT between S-C
13PO
13S and oil gradually decreased and reached an order of 1×10
−2 mN/m as shown in
Fig. 8a. The excellent interfacial activity of S-C
13PO
13S is due to its long PO chain which has a size compatible with its hydrophilic group
[15]. He et al.
[26] proposed that the long PO chain of the extended surfactant adjusts the hydrophobic group size to achieve an ultra-low IFT by curling into a spiral at the oil-water interface uniformly. In
Fig. 8b, the air-liquid-solid contact angle of S-C
13PO
13S solution on the quartz surface is significantly smaller than that of distilled water. This indicates that S-C
13PO
13S reduces the surface tension significantly
[27]. The oil-liquid-solid contact angles indicate that S-C
13PO
13S molecules are adsorbed at the oil-water interface, and the anionic heads towards the aqueous phase increase the negative charge density at the interface significantly (
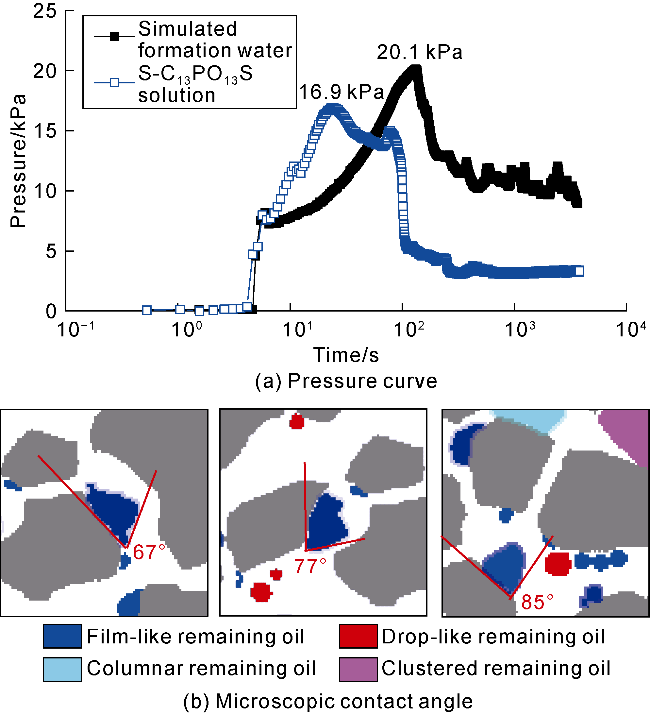
Fig. 8c). These negative charges generate a larger electrostatic separation pressure near the three- phase point and increase the oil-liquid-solid contact angle rapidly to 125°, which is conductive to oil film separation
[5]. Zeta potential measurement at the oil-water interface shows −15.2 mV for simulated formation water and −58.6 mV for S-C
13PO
13S solution, confirming that S-C
13PO
13S enhances electrostatic interaction significantly.
Fig. 8d shows that S-C
13PO
13S emulsion cannot provide water separation within 60 min. This is due to the helical structure in the molecule with stored energy that can deform in response to interface disturbances, thus increasing the strength of the interfacial film and improving the emulsion stability (
Fig. 8e). Tagavifar et al.
[28] reported the kinetic behavior of C
13-PO
13-SO
4 in micro-emulsion that is formed intermediately as C
13-PO
13-SO
4 solution contacts with oil.