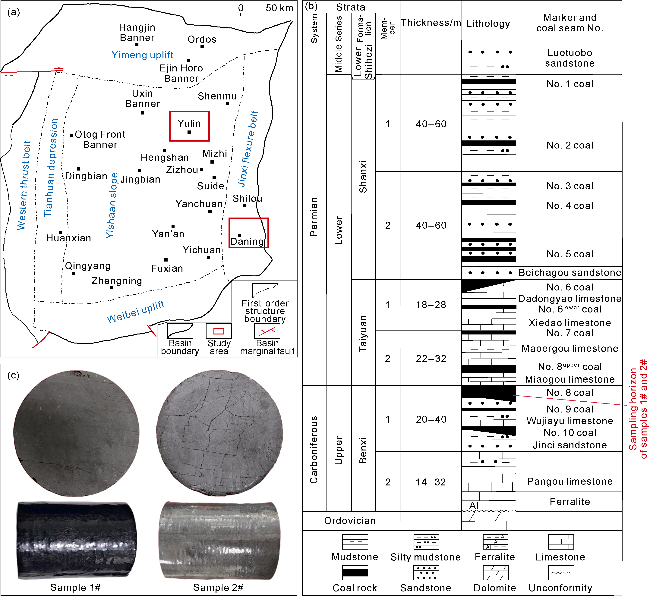
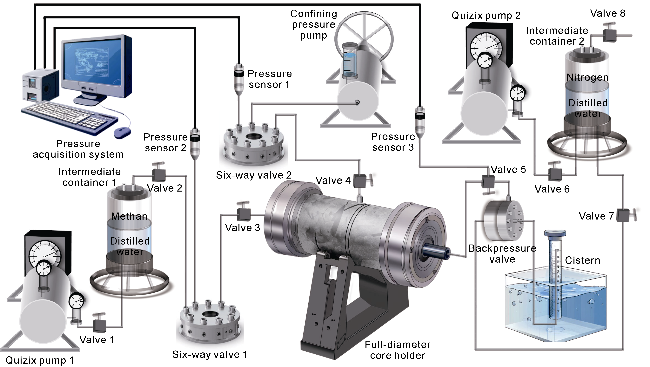
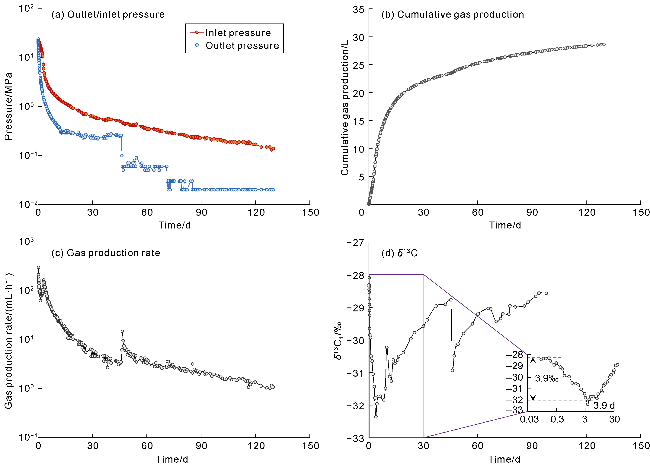
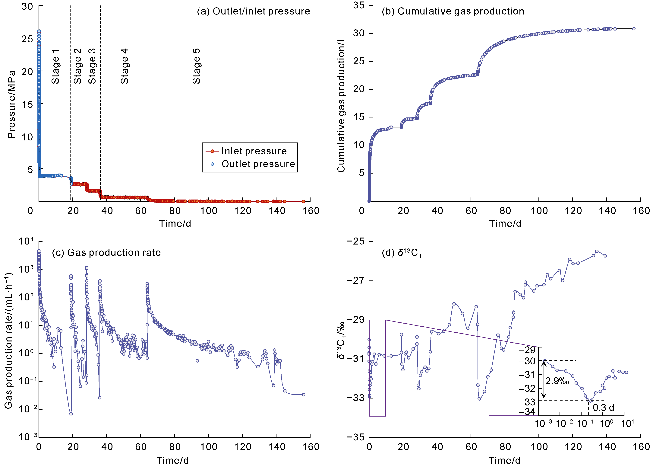
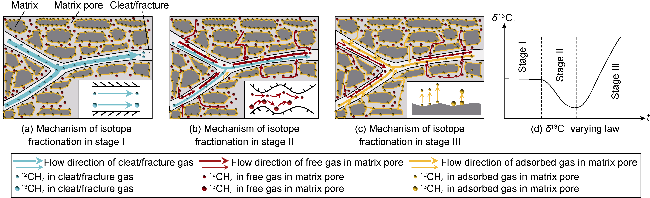
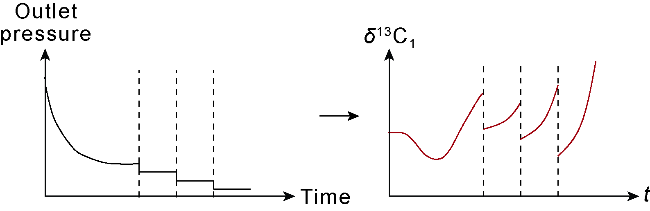
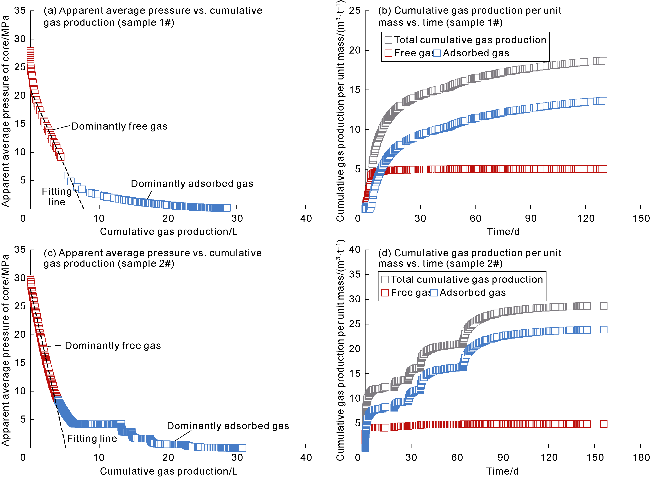
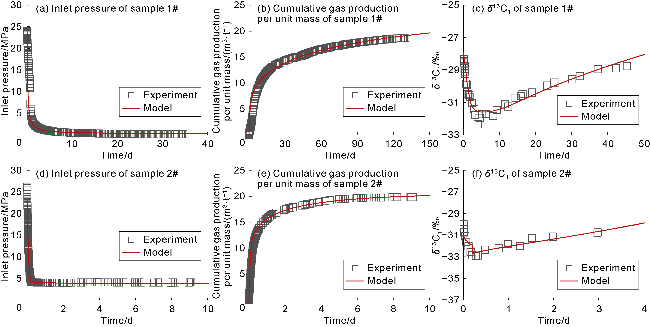
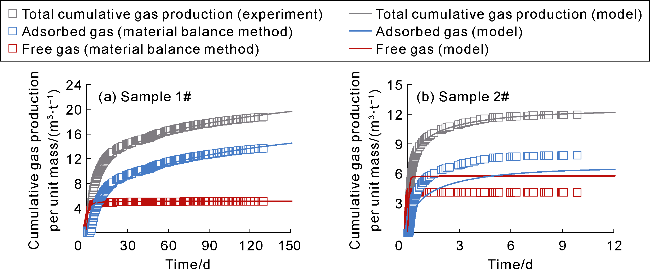
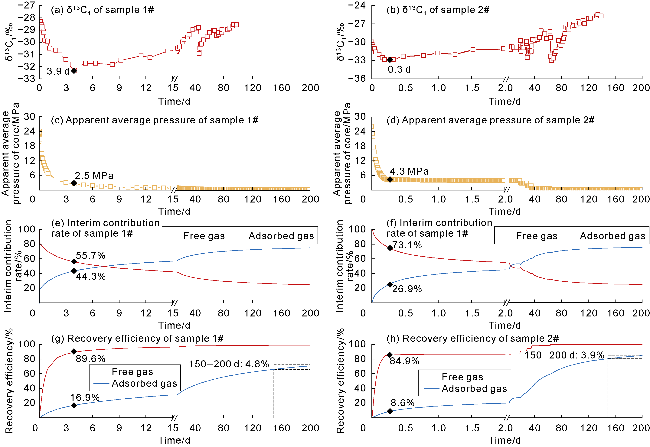
With gas production from the cores, the pressure inside the cores decreases, and both samples show a decrease in the interim contribution rate of free gas and an increase in the interim contribution rate of adsorbed gas. The inflection point (P
2) between stages II and III of isotopic fractionation in sample 1# occurs around 3.9 d (
Fig. 10a), corresponding to the interim contribution rate of free gas being 55.7% (
Fig. 10e). The P
2 point of sample 2# occurs around 0.3 d (
Fig. 10b), corresponding to the interim contribution rate of free gas being 73.1% (
Fig. 10f). Notably, the two coal-rock samples have different degrees of cleat/fracture development, but both exhibit the dominance of free gas (interim contribution rate greater than 50%) in the stage II of isotopic fractionation. The recovery efficiency of free gas corresponding to P
2 of samples 1# and 2# are 89.6% and 84.9%, and that of adsorbed gas are 16.9% and 8.6%, respectively (
Fig. 10g,
10h). This indicates that most of the free gas has been produced in stages I and II of isotopic fractionation, with the recovery efficiency greater than 80%, but there is still a large amount of adsorbed gas remaining, which is the reason for the high initial production rate and long stable production period of deep coal-rock gas wells. In the initial production stage (before P
2), the apparent average pressure of cores declines rapidly (
Fig. 10c,
10d), and the recovery efficiency of adsorbed/free gas increases significantly, with the recovery efficiency of free gas growing at a rate greater than that of adsorbed gas (
Fig. 10g,
10h). In the middle and late stages (after P
2), the pressure declines slowly (
Fig. 10c,
10d), and the recovery efficiency of free gas grows slowly, at a rate lower than that of adsorbed gas (
Fig. 10g,
10h). The staged pressure control production method effectively increases the recovery efficiency of adsorbed/free gas, especially free gas that is difficult to produce. When the pressure drops to the atmospheric level (after 150 d), the recovery efficiency of free gas approaches 100%. The producibility of adsorbed gas depends on the desorption capacity of the rock. Although the permeability of sample 2# is higher than that of sample 1#, during the production period of 150 d to 200 d, the interim recovery efficiency of adsorbed gas in sample 2# (3.9%) is lower than that in sample 1# (4.8%). Sample 2# has higher "vitrinite + inertinite" content and lower Langmuir pressure, which are 96.4% and 1.82 MPa, respectively, in contrast to the values (93.6% and 2.31 MPa) of sample 1#. Generally, the adsorption capacity of vitrinite and inertinite to methane is higher than that of exinite. The lower the Langmuir pressure, the greater the adsorption capacity of rocks is. The measured "vitrinite + inertinite" content and the Langmuir pressure determined by history matching both indicate that sample 2# has greater adsorption capacity, so its adsorbed gas production capacity in the late stage is weaker than that of sample 1#.